Tag: bias in drug studies
How Do We Know When a Treatment Works? A Primer on...
Discussing informed consent, risk/benefit ratios, and the many sources of bias in clinical trials for drugs, in order to help the layperson better understand the research.
Mental Health Survival Kit, Chapter 2: Is Psychiatry Evidence Based? (Part...
Virtually every single placebo-controlled drug trial in psychiatry is flawed, systematic reviews of trials are also flawed, and guidelines are flawed. Even the drug approval process is flawed.
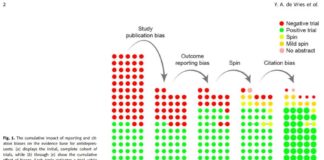
Publication Bias Inflates Perceived Efficacy of Depression Treatments, Study Finds
Researchers report the cumulative effects of major biases on the apparent efficacy of antidepressant and psychotherapy treatments.
Poor Evidence and Substantial Bias in Ritalin Studies
The authors of a large scale well-conducted systematic review of methylphenidate, also known as Ritalin, conclude that there is a lack of quality evidence for the drug’s effectiveness. Their research also revealed that Ritalin can cause sleep problems and decreased appetite in children.