Tag: bias in industry trials
Michael Hengartner – Evidence-biased Antidepressant Prescription
We talk with Dr. Michael Hengartner about his new book which addresses the overprescribing of antidepressant drugs and critically examines the scientific evidence on their efficacy and safety.
How Do We Know When a Treatment Works? A Primer on...
Discussing informed consent, risk/benefit ratios, and the many sources of bias in clinical trials for drugs, in order to help the layperson better understand the research.
Mental Health Survival Kit, Chapter 2: Is Psychiatry Evidence Based? (Part...
Virtually every single placebo-controlled drug trial in psychiatry is flawed, systematic reviews of trials are also flawed, and guidelines are flawed. Even the drug approval process is flawed.
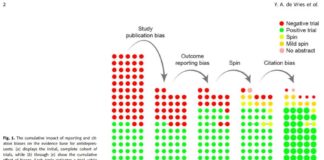
Publication Bias Inflates Perceived Efficacy of Depression Treatments, Study Finds
Researchers report the cumulative effects of major biases on the apparent efficacy of antidepressant and psychotherapy treatments.
“NIH-Funded Trials Dip, Industry Trials on the Rise”
"Every year since 2006 in the U.S., the amount of new medical research in humans that’s funded by the National Institutes of Health (NIH) has gone down, while the number of industry-funded trials has gone up, a new study shows.”