The Cochrane Collaboration was established in 1993 as an idealistic grassroots organisation with the aim of publishing updated systematic reviews of the benefits and harms of interventions in healthcare. It started in Oxford in the UK, spread rapidly throughout the world, and became very successful.
Unfortunately, like most successful grassroots organisations, it came to suffer from institutional corruption early on. This moral decline accelerated when a new CEO, journalist Mark Wilson, was appointed in 2012. I co-founded Cochrane in 1993 and have described its moral and scientific downfall in two books. This sad story will also be told in a documentary and in my upcoming podcast series, Broken Medical Science.
 Cochrane reviews are published in the Cochrane Library, once at the forefront of science, but now an anachronism, as is Cochrane’s marketing logo, “Trusted evidence.” This is particularly true with Cochrane reviews of psychiatric treatments; they can’t be trusted.
Cochrane reviews are published in the Cochrane Library, once at the forefront of science, but now an anachronism, as is Cochrane’s marketing logo, “Trusted evidence.” This is particularly true with Cochrane reviews of psychiatric treatments; they can’t be trusted.
Today, Cochrane is keen to protect guild interests and to comply with official views from governments even when they contradict the evidence in Cochrane reviews, as illustrated by its recent review of face masks. Cochrane is too close to industry, and Cochrane does not care much about financial conflicts of interest for its review authors.
When I was elected to the Cochrane Governing Board in 2017, I called for a strengthening of our conflicts of interest policy. This was agreed to, and I rewrote the policy in an afternoon. But the Cochrane leadership ignored it and spent over two years consulting with Cochrane members before its “strengthened” policy came out. Today, 33% of new review authors can be on company payroll. Before, it was 50%. Semmelweis would not have liked this “revolutionary” new policy. He never told doctors to reduce contamination by washing one hand. He told them to wash both hands.
The game for Cochrane seems to be over. Cochrane’s CEO Mark Wilson showed no interest at all when, a decade ago, Tom Jefferson demonstrated that Cochrane reviews of drugs against influenza based on published trial reports are totally misleading. I demonstrated that this was also the case for psychiatric drugs. Wilson suddenly left Cochrane in the middle of a month, in April 2021, with no explanation, seven days before the UK funder announced that a major budget cut was likely. The current leadership shows no interest either in this vitally important issue of whether its reviews are misleading.
In April 2021, Professor Ken Stein, Director of the Evidence Synthesis Programme at the UK National Institute for Health and Care Research (NIHCR), the major funder of Cochrane, criticised Cochrane at a webinar for much the same reasons as I had when I joined the Cochrane Governing Board. He said the writing had been on the wall for eight years, which was exactly the period when Mark Wilson ruled the organisation and destroyed it.
About the failing scientific integrity, Stein noted that, “This is a point raised by people in the Collaboration to ensure that garbage does not go into the reviews; otherwise, your reviews will be garbage.” And as Tom Jefferson said in an interview in 2018, such reviews are published with a nice little Cochrane logo on the garbage.
There were, at the start of 2023, 52 Cochrane review groups in the world, together covering all aspects of healthcare. However, in March 2023, all 24 with a base in the UK lost their government funding. Apart from four groups that have other funding sources, they have all now closed down. In July 2023, the NIHCR announced that it will end its contract with the UK Cochrane Centre in March 2024, a year before it was due to expire. The reason? Inefficiency and lack of relevance of the Cochrane reviews.
Cochrane has become a self-serving institution, a hugely ineffective bureaucracy, which is succumbing under its own weight, as illustrated by the size of Cochrane reviews. The median size was 26 pages in 2001, 41 in 2012 and 79 in 2023. A few are so big that they correspond to several books.
If you have a collection of, say, 30 drug trials that are all deeply flawed, which is typically the case in psychiatry, you are not allowed to say this in a short review. Instead, you are required to write a very long review with details about every single trial even though it is impossible to conclude anything based on them. Yet, even given this meticulous attention to detail, most Cochrane authors and editors pay far too little attention to all the flaws in the trials and draw conclusions about drug benefits and a lack of harms that are unjustified.
Cochrane reviews of psychiatric drugs should generally be ignored. Virtually all of them are based on untrustworthy published trial reports, rather than on the lengthy clinical study reports the manufacturers have submitted to drug agencies to get approval for their drugs. In particular, reviews of placebo-controlled trials are a garbage in, garbage out exercise.
Cochrane editorial misconduct in relation to our review of safe withdrawal of depression drugs
In 2016, I contacted psychiatrist Rachel Churchill, editor of the Cochrane Common Mental Disorders group, who showed great interest in my proposal to do a review on safe withdrawal of depression drugs. The story of how Cochrane treated this proposal provides a case study of how Cochrane protects guild interests.
This was a very important review. Hundreds of millions of patients have become dependent on depression drugs, just like patients become dependent on benzodiazepines. The withdrawal symptoms are very similar; about half have difficulty stopping; and doctors don’t know how to taper the drugs safely and therefore usually just renew the prescriptions, for many years on end.
But we quickly faced a roadblock. The Cochrane group was extremely slow to respond and imposed ever increasing demands to our protocol. At the same time, they negotiated secretly with another author group on the same issue. This violated two of the ten Cochrane key principles: Collaboration, which involves open and transparent communication and decision-making, and avoiding duplication of effort, which is about avoiding that two reviews cover more or less the same ground.
The Cochrane group rejected our protocol two years and four months after we first submitted it, while it accepted the other authors’ protocol and published a very embarrassing Cochrane review, which is full of misleading statements and marketing messages of great value for the drug industry but irrelevant for doctors and patients (see below).
On 28 March 2023, I sent a complaint to Karla Soares-Weiser, Cochrane’s Editor-in-Chief, about editorial misconduct in an open letter. Later, I also sent a complaint to Cochrane’s current CEO, Catherine Spencer. I asked some simple questions, which they refused to answer. I have described the bizarre interactions I had with the Cochrane leadership elsewhere. Briefly, they beat about the bush, just like the drug industry does when you have a really good case against them. I couldn’t see the difference. As Jefferson once said in relation to Roche’s unwillingness to provide the data from its unpublished trials of Tamiflu for influenza, If you have nothing to hide, then hide nothing. Cochrane hid everything in relation to our review.
It also turned out that Cochrane has no mechanism for handling allegations of editorial misconduct in an impartial manner, something all reputable journals have. My translation of the message I got from Cochrane’s CEO was this: “We don’t give a damn. We are beyond reproach.”
Cochrane abused the peer review process to the extreme
Our protocol for reviewing drug withdrawal studies was rejected in November 2019. However, in February 2020, a similar protocol about withdrawing depression drugs was published in the Cochrane Library. It takes a very long time to get a Cochrane protocol approved and published, which means that this project must have been underway secretly for many months while the review group increased their demands regarding our protocol, which I see as an effort to wear us out without their being seen as uncooperative.
Cochrane abused the peer review process to the extreme. Four editors and three peer reviewers provided individual comments, and, including our replies to earlier comments, the peer review document took up 12,044 words, seven times the number of words in our original protocol. My co-author wrote to me that our review was quite simple and that we just wanted to help people who wished to come off their drugs, but we weren’t allowed to do so. He asked: “What kind of world is this?”
The 8th peer review was as long as our protocol. It was clear to us that the final reviewer’s mission was to protect psychiatry’s guild interests, providing an excuse for the editor to reject our protocol. This was done by denying a long array of scientific facts; by using strawman arguments accusing us of things we had never claimed; and by requesting us to discuss issues that were totally irrelevant for our review and to insert text that was blatantly wrong.
In contrast to the 7 previous reviewers, this one was anonymous. We asked for the identity of the “hangman,” but this was not granted. I noted that very few changes to the protocol were needed and submitted a new version and a rebuttal with solid scientific arguments against the many errors and misguided opinions in the peer review, but this didn’t matter the slightest bit for Cochrane.
The 8th peer review is one of the worst I have ever seen. I published a detailed account of it four years ago and shall only summarise the main issues here.
The reviewer accused us of “painting a picture” about antidepressants being “bad medications” to be avoided, which did not represent the scientific consensus, and wanted us to “Start with a statement as to why antidepressants are considered by the scientific community to be beneficial … in treating a broad range of highly disabling and debilitating mental health problems.” The reviewer found it “unscientific, and unacceptable in the context of the current evidence base” that we had not mentioned the beneficial effects. We responded that our review was not an advertisement for the drugs and that it was not relevant to discuss their effect in a review about stopping using them.
The reviewer believed that a chemical imbalance in the brain was the cause of depression and wanted us to write about this. We responded that our review was not the place for such discussions and that the hypothesis of a lack of serotonin being the cause of depression had been discredited by many convincing studies.
We were asked to explain the concept of ongoing prophylactic antidepressant treatment, “a well-accepted clinical strategy,” but this was outside the scope of our review. Furthermore, the randomised trials comparing maintenance therapy with drug withdrawal are seriously flawed by their cold turkey design. Some of the withdrawal symptoms in the placebo group mimic depression.
The reviewer wrote that we conflated relapse with withdrawal symptoms, which wasn’t true. But many psychiatrists do, which is a major reason why many patients are treated for decades or for life.
The reviewer argued that most people who had taken antidepressants for extended periods could stop safely without either rebound of the disease or withdrawal symptoms. This is untrue, which we had documented and referenced in our protocol. The UK Royal College of Psychiatrists reported in 2012 that 63% of 817 people who had stopped taking depression pills experienced withdrawal symptoms.
The reviewer wanted us to remove this sentence: “the patients’ condition is best described as drug dependence” arguing, with reference to the DSM-IV drug dependence criteria, that it is an unreasonable misappropriation of a term. We responded: “The official definitions of dependence are ridiculous and self-serving, in addition to serving the drug companies that have benefitted hugely from the false perception that only benzodiazepines cause dependence, not the SSRIs. Craving larger and larger doses as a criterion for dependence is absurd, as it means that no one who smokes 20 cigarettes every day is dependent on smoking cigarettes!”
The published Cochrane review of withdrawal was of poor quality and filled with industry-like marketing messages
While we were being blocked from conducting a drug withdrawal review for Cochrane, the other group that had submitted a protocol for doing so was given the green light to proceed, and in 2021 Cochrane published it. The review was restricted to adults with depression or anxiety, which is irrational. The drugs are used for many conditions, and the withdrawal symptoms are not dependent on why the drugs are prescribed or the age of the patient. Moreover, the review did not include trials comparing different withdrawal strategies, which we did, whereas it included many flawed studies comparing abrupt discontinuation (cold turkey design) with continuation. We only included trials that had at least one treatment arm that aimed to help patients withdraw from a depression drug.
The published Cochrane review included 33 studies (4995 participants). In our review, which we first published as a preprint (likely to come out in a medical journal soon), we included 13 studies (2085 participants). The Cochrane review is 209 pages (110,770 words), the length of a full book, 23 times as long as our review of 9 pages.
I studied the Background section in the Cochrane review, which, with its 4239 words, is longer than most scientific papers. It is full of irrelevant marketing messages and misleading statements, which I noted in my complaint to Cochrane about editorial misconduct.
It states that “Maintenance treatment is provided to prevent recurrence.” There is no mention that all maintenance trials are deeply flawed and that the patients’ withdrawal symptoms are erroneously interpreted as a relapse of the depression.
“Antidepressants have been shown to be efficacious in adults … (Cipriani 2018) … between seven and eight people needed to be treated with an SSRI … for one person to experience improvement.”
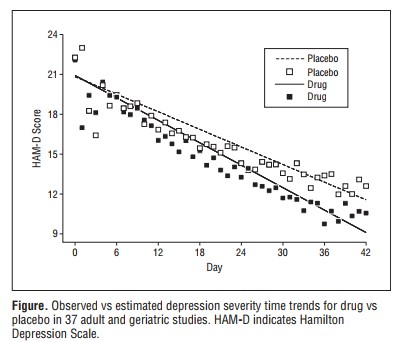
These praises of drug benefits are invalid. The effect on the Hamilton scale, a difference of 2 over placebo, also in Cipriani 2018 (an effect size of 0.30 corresponds to a Hamilton difference of 2.3), is far below what is clinically relevant, as the least difference on the Hamilton scale that can be detected is about 5-6.
I have shown why the number needed to treat (NNT) with a psychiatric drug to benefit one patient is largely an illusion. One reason is that more patients are harmed than those who benefit. When patients decide whether it is worthwhile to continue in a trial, they make a judgement on whether the benefits they perceive exceed the harms. Based on clinical study reports we obtained from drug regulators, we found that 12% more patients drop out on a depression pill than on placebo (P < 0.00001). Thus, the patients consider placebo more useful than a depression drug, which means there cannot be an NNT, only a number needed to harm (NNH). Our meta-analysis showed that this number is about 25. The published trial reports do not reveal this devastating finding.
When the top among UK psychiatrists in 2014 tried to convince their readers that depression pills are highly effective, they mentioned an impressive effect on recurrence, with an NNT of around three. But these trials did not assess recurrence but withdrawal symptoms in the placebo group. As only two patients are needed to get one with withdrawal symptoms when a drug is stopped, there cannot exist an NNT to prevent recurrence, only an NNH, which is two.
Since depression pills harm the sex life in about half the patients, the NNH is two. Thus, by not using depression pills, we will preserve the normal sex life in one out of every two patients we do not treat.
The published Cochrane review is unbalanced. It gives precise but misleading estimates of the benefit in the form of NNT but does not offer similar estimates for the most serious harms. This goes against the very ethos of Cochrane, which always was to focus similarly on the benefits and harms of interventions. The Cochrane review mentions the worst harm, suicidality and suicide, in many places, but it does not say that depression drugs double the suicide risk, both in children and adults.
The Cochrane review states that “evidence suggests that continuation of antidepressant treatment is effective, as it reduces risk of relapse and recurrence by 50% to 70%.” The authors should not have propagated this horrible misinformation, but rather should have stated that there is no reliable evidence that the drugs reduce recurrence.
The review continues: “The effect of most antidepressants fully develops after some weeks, indicating that neurophysiological changes in brain tissue … are necessary for improvement in depressive symptoms.” This neuro mumbo jumbo is also highly misleading. One cannot say that the effect fully develops after some weeks when there is a very gradual and slow separation of the Hamilton scores on drug and on placebo and when the effect even after seven weeks is so small that it is not clinically relevant:
The authors note that “suggesting that a single biochemical deficiency is the cause of depression and that antidepressants work by correcting chemical deficiency is not correct.” Interesting that they were allowed to say this. We were heavily criticised by the hangman for having written that the hypothesis about a chemical imbalance in the brain being the cause of depression had been discredited.
One of the main aims in establishing the Cochrane Collaboration in 1993 was to assist patients in their decision making. However, the whole Background section is about what doctors think and the review is highly paternalistic. There is no mention that many patients want to come off the drugs, which should have been the key motivation for the authors to do their review.
There is no mention in the Background section that the tapering should be hyperbolic, which is essential if one wishes to minimise withdrawal symptoms, whereas the authors quote a NICE guideline from 2009 without criticising it for recommending a fast, non-hyperbolic tapering that is outright dangerous, as it can cause akathisia, suicide and violence. When we started Cochrane in 1993, we were willing to criticise the authorities. The current leadership wants to please the authorities and the drug industry, which this Cochrane review demonstrates.
The abstract of the Cochrane review is 915 words, four times as long as ours of 236 words. But length is not a substitute for quality. The Cochrane abstract states that “We cannot make any firm conclusions about effects and safety of the approaches studied to date” and that “Future studies should report key outcomes such as successful discontinuation rate.”
Our little abstract is far more informative. We included fewer but more relevant studies and found a median successful discontinuation rate of 50%. More importantly, we noted:
“A meta-regression showed that the length of taper was highly predictive for the risk of relapse (P = 0.00001). All the studies we reviewed confounded withdrawal symptoms with relapse; did not use hyperbolic tapering; withdrew the depression drug too fast in a linear fashion; and stopped it entirely when receptor occupancy was still high.” We concluded that “The true proportion of patients on depression drugs who can stop safely without relapse is likely considerably higher than the 50% we found.”
The lengthy Cochrane abstract did not mention any of these essential issues.
Cochrane is in deep trouble
Currently, faced with an ongoing crisis caused by multiple missteps, Cochrane’s Editor-in-Chief, Karla Soares-Weiser, has hired the pricey consulting firm Envoy to address scientists’ concerns about her lack of transparency, leadership, and communication skills.
The crisis came to a head in March, when Soares-Weiser rushed out an apology even though there was nothing to apologize for, which undermined Jefferson’s and colleagues’ Cochrane review that showed that masks did not reduce respiratory viral infection. Soares-Weiser even claimed, totally falsely, that “the review is not able to address the question of whether mask-wearing itself reduces people’s risk of contracting or spreading respiratory viruses.” This was one of the aims of the review!
When investigative journalist Paul D Thacker asked for insight into how Cochrane had handled his request for comment and answers in relation to this scandal, he received heavy redacted documents:

So much for Cochrane transparency, which appears in the first of Cochrane’s 10 key principles: “Collaboration by fostering global co-operation, teamwork, and open and transparent communication and decision-making.” What is the difference to the drug industry?
Thacker called his article: “Cochrane: world’s preeminent medical information resource goes into tailspin.” It should come as no surprise that many people who were previously very supportive of Cochrane now see it as a slowly dying organisation.












Thank you, Dr. Peter, for speaking the truth about how one should actually be properly weaned from the antidepressants. Since our current “mental health” industries, and the mainstream medical and pharmaceutical industries, are now largely claiming ignorance of the common adverse and withdrawal effects of their antidepressants. Not to mention, complete and total ignorance into how to safely wean people off the psych neurotoxins.
Including the fact that both the antidepressants and antipsychotics (both anticholinergic drugs) can create “psychosis” and “hallucinations,” via anticholinergic toxidrome poisonings.
https://en.wikipedia.org/wiki/Toxidrome
Information all the MDs, including the psychiatrists, were all taught in med school. So I have no idea (other than insane greed, and abuse of power) why too many of the mainstream doctors are inappropriately prescribing, and lying about the potential common adverse and withdrawal symptoms of their neurotoxic psych drugs.
Thank you so much for all the truth you tell, Dr. Peter. And maybe we are seeing, or will some day see, God’s will be done, and a better world some day. But I do know, “the truth will set you free.” So thank you to all the truth tellers, including you, Dr. Peter.
And with the Cochrane group’s seeming chosen destruction of their own credibility, maybe we’re seeing a little justice from God? And the honest truth tellers may have a chance in winning some day. Let’s hope and pray for that, at least.
Report comment
One thing that on second look of the graph is pretty obvious to me is that one VERY effective intervention for depression, i.e. it decreases the HAM-D score from around above 21pts to around 12pts, that is a 9pts difference of the intervention at 6 weeks (or 1.5pts per week decrease!), which is at least 3 times the diference at the end of 6 weeks between the placebo and the drug, is: just join a controlled study! in the placebo group!.
Without taking any drug. Weirder and more counterintuitive when, if that were the case, the placebo group was taking drugS before being allocated to placebo!.
I am not recommending stopping cold turkey, on own’s own or on current guidelines, btw, which to me is in part the point of this PG piece.
But that sounds a lot like going to psychotherapy without taking any drugs: my HAM-D score will decrease by being in a place/situation under conditions of this/that controlled study.
And I have to keep reminding myself that if I extend the straight curve from the placebo group, ceteris paribus, all else being equal, at 12 weeks my average score will be 3pts if I started with 21, and 2 weeks later, i.e. at 14 weeks, my average HAM-D score will be ZERO.
Just by joinning this/that controlled study in the placebo group. Which to me speaks volumes of using a straight line for non linear data, to say the least, among other things.
Granted achieving ZERO in the HAM-D score might not be achievable for anyone or everyone, but that’s what the stright curve, the line in popular terms would suggest.
The minimal HAM-D score in the population might actually be 10 or 11pts, maybe more, maybe less, I have no idea.
Maybe as a Freudian slip of the researchers the minimal HAM-D score in the population, normal population, ALL the individuals above 18yrs, etc., might actually be around 8pts, who knows, and that’s why the bottom of the Y-axis is just below 9pts…
Report comment
I think this piece by PG is of historical significance.
I doubt historians are going to take much interest in writing this as part of history now.
Since according to a lecture of historiography from the London School of Economics of 1 or 2yrs ago, and my informal readings in history, historians are more focused in studying what was said about the past, rather than documenting for future historians the present.
Even if it’s from second hand.
And there are biographical exceptions of which Enrique Krauze is one outstanding among them. There are others but the stack is loaded against analysis of current events in historiography, not in providing historical analysis. i.e. what historians in part do.
And even in that last case, there are not many outlets even in the mainstream to provide a megaphone for them. But that is my opinion.
For me even History Extra picks just themes of geopolitical importance for the most part. Some racism, human rights, women’s rights, etc., but very few if at all, about what PG narrates here. In a historical context of course.
Unlike the march of the 10,000s model of historiography. Even Herodotus and his mythical ants recorded events from his own time for future historians to dwelve into it. And a lot of what Herodotus recorded was from second hand…
I’d love to be proven wrong on the fights of PG to be picked up by current historians who could provide historical analysis to his deep and wide view of some of the ills of current medicine, and particularly not exclusively psychiatry. And I’ll faint if they actually write history about it…
Report comment
The graph in this PG article seems odd in many ways:
It lacks error bars, there is no way to know where in the HAM-D score 95% of the patients fall along the curves depicted. No idea of the spread of scores.
It is fitted to a straight line when it seems to fit better a “hammock” curve, belly downwards and to the left. Not a straight line curve. And even then, the dispersion of average HAM-D scores at the begining and at the end look like an hoour-glass, not even a hammock.
If I draw a line joining the dots of each curve I see a severely zigzaging line both for the placebo and the drug intervention at the begining. A thing recomended to everyone who looks at graphs: draw the line to join the dots.
The last 6 days of the drug in the graph seem to flatten suggesting no further improvement. Less obvious in the placebo, where it even ligthly suggest the scores were about to go up. That in the drug group is possible, but without the spreads is difficult to know.
Another thing recommended to people looking at graphs is to consider each part of the graph independently and not get hooked up by the fitting curve, which in this case is a straight line, which is IMO misleading.
Going on a limb, I would speculate the graph is of patients that were switched from previous medications to the current drug or placebo, and for the first week, it was a roller coaster for the placebo and the drug intervention. That suggests to me the switch, if it happened, exposed patients to at least emotional instability reflected in the HAM-D score variability. That sounds bad to me, given they were research subjects.
I wonder if the informed consent warned them of that explictly?
And in practical terms such as employment, homelessness, agression, self harm, incarceration, etc., what are the ourcomes for such HAM-D scores oscillation on a daily basis!?
Without the spreads I can only be conservative about it, might it be some patients went below or above, somehow the HAM-D scale during the first 6 days?. Not imputing, just arguing for the need of the error bars, the spread of the individual scores, and not just reporting the average, because that spread and in this case the oscillations might have real consequences that trascend psychiatry to spill in the real world.
Report comment
You’re ignoring some important facts. Mental illnesses including depression have HIGH RATES of homozygous MTHFR mutations. In other words, you’re dealing with people who have high histamine, high homocysteine, inability to metabolize Folic Acid, low Vit B12 and Vit D.
Then you give them antidepressants that do what? Lower calcium, Vitamin D and some of them increase histamine….a great reason for non-response. Antidepressants are also known to cause coagulation problems and it just so happens so do homozygous C677T MTHFR mutations.
If you then discontinue antidepressants, you will be causing further methylation dysregulation and the symptoms you’ll see will be the same as those caused by the methylation dysregulation caused by homozygous MTHFR mutations….which is exactly what the people in the antidepressant withdrawal groups are having.
The ones who have been tested all seem to have homozygous C677T MTHFR mutations.
Report comment
I don’t have much to say about the veracity of Cochrane as an institution and the authenticity of the work it does, and I never talk crap or reveal personal information about any of my doctors (I’ll make an exception in this case). I respect their privacy as much as they respect mine. My dentists and I sometimes casually chat about our personal lives but I would not repeat it here or anywhere else in public without their permission. This is common social courtesy and it is a matter of mutual trust between friends, family members, colleagues, doctors-patients etc. The problem is, when you’re a psychiatrist and your words are considered by families like they’ve been uttered by God and you overpower the words of the person who is actually suffering and it prevents them from getting justice, it becomes a serious problem. I can’t help it if it spoils their reputation because they did not bother when they treated me like some mannerless street urchin without even bothering to understand what I was going through and just kept assassinating my character and disease-mongering. Sorry, this is going to be a very big “rant” and unlike any of my other posts here (which are on topic). To someone else, all this may be “anecdotal evidence”, but this is my life. It is very personal, but I have almost nowhere else to express myself.
I come from a much poorer, developing country: India. Though I did not grow up poor, my paternal family is from a very small, rustic, conservative, underdeveloped, rural area. My maternal family is a bit different. As a result of this difference, I was the first person out of nearly 30 people in my paternal family, who grew up in a city, who could speak fluent English, had some degree of liberal values etc. When you’re someone from my demographic here, it tends to attract a lot of resentment, disdain and even jealousy, be it from society in general or even within your own family (you can see this mentality on the Indian side of social media as well). My Urologist “father” was such was a resentful person too. I will call him RB.
The (now ex) South Asian Director of Cochrane Collaboration was the first and pretty much the last psychiatrist I had. I will just refer to him as Mr. SAD (for South Asian Director). I met him when I was 16 through a family member who already knew him (this prior “history” is where my headache started from). I did not know what Cochrane was at the time, except I knew the word because of a small sticker pasted onto his office door.
At the time of first contact with Mr. SAD, I was utterly terrified of RB who used to, and still does, behave like an absolute psychopath: brutal gaslighting, pathological lying, philandering, constant degrading and insults without end, and overall just aggressive crude rustic behaviour. It has seriously hurt my life, mind and academics. I used to be so terrified of RB that in his presence I could not speak and my head used to bow down. It was that way for years. I was reading an article on gaslighting a few days back and it describes his behaviour so perfectly that I was shocked regarding its accuracy. Clearly, there are people like him all over the world. Hell, I’m in my 30s now and his behaviour still haunts me and he is more than simple flesh-and-blood man in my mind. I was depressed, fearful and I had all sorts of obsessive-compulsive symptoms. I was so screwed up at the time that if you told me “hey registeredforthissite, if I hit you on the head with this brick, it will hurt for a while but then you’ll be all fine”, I’d have said “yes, please, anything to end the pain”. I knew absolutely nothing about Psychiatry. I did know physical medicine and surgical departments pretty well though because I used to hang around them all the time.
I don’t want to assassinate Mr. SAD’s character, because he is fundamentally a good man, a good family man, and has a moral centre. Also, he is very accomplished with a large number of publications. He’s nothing like my own father, but is unfortunately a “goodie good mommy’s boy” who has no idea what it is like to grow up in an abusive family. Basically, what I would have been, had I not grown up in the hellhole I did. Reading about this stuff in textbooks, having clients speak about it and actually experiencing it is very different.
However, just like a large chunk of other psychiatrists, Mr. SAD would refuse to listen to anything said against his field and would rather disease-monger and invalidate than admit any shortcomings. His colleagues were actually worse and I had to beg them like a dog to get him back because he was the first psychiatry “doctor” I ever had and he labelled me as “bipolar” for mania caused by a drug called Sertraline when I was 16 (I do not have a problem with the drug causing mania, nor being prescribed it, I needed something to alleviate my suffering, but that term itself became a source of incessant gaslighting). His junior doctors in the interim when he was unavailable were denying that SSRIs had anything to do with causing mania, that it was chance correlation that I happened to experience it when I was on them (which is rubbish) and that I have a “family history”. This “family history” consists of just one person, a second degree relative. I asked her too and she said “it is the antidepressants that used to get me high/manic and my doctor said it was medication induced”. She was prescribed these drugs by Mr. SAD too, when he was younger and down lower in the medical hierarchy. The only person who could verify that it was medication induced in me was him, so I needed him back because his juniors in the interim were trying to con me out of the truth. Labelling people with a term that’s associated with occurrences like becoming psychotic for no reason at all for a medication induced effect is digusting.
Mr. SAD lost his dad as a kid. As a result, earlier in my youth, Mr. SAD’s “advice” was “you are secretely proud of your father and longing for his affection”, “if you hurt your father you will only hurt yourself”, “fathers and sons can’t have the relationship they must have till they go through what fathers and sons go through”. I was young and went along with it. I waited for years and none of it came true. In fact, RB just kept deteriorating over time. I knew he would but I had no power to change the opinions of the adults who had power over me.
All the hell of my childhood from RB not being enough, in 2014-2015, RB was having an affair with his housemaid. It was in the same house in which I grew up in my mid-teens. In order to cover it up, he was gaslighting me brutally, threatening to get me institutionalised in a psych ward, and put all sorts of horrible false allegations on me. His maid used to enjoy all the lavishes he would provide, roam around in his cars, and he basically maintained her as a concubine/mistress. I was mortified seeing these scenes. As usual, no matter how hurtful his behaviour was, he would continue it shamelessly. When the affair became a bit more public, in order to save himself, RB got the maid married off to another man. The maid and her husband have a son. Even in 2023, RB lives in our old house along with this maid, her husband and her son. They have stuck around and created a nexus in order to siphon off wealth and properties from him. I cannot even go to the very area in which I grew up because of these people and I am afraid that they may endanger my life.
In 2014-15, I used to walk around like a vagabond on foot everywhere, do a menial job and I was totally traumatised as a result of all this. RB and his maid, basically tried turning me into a non-entity. They tried wiping out my existence from the city in which I grew up, cutting off the connections that I am actually that man’s child. RB would introduce his maid (that he’s banging) as his daughter everywhere. If any of the few people who remember me asked about me, I’d just be described as a “schizophrenic case” or a mad person. RB is also supported by a family of over half a dozen of his siblings and over a dozen of those siblings’ children. They get reputation boosts by throwing around his name, and free medical treatment and lodging when required. He’s nice to them. Throughout my childhood, I’d avoid them because they backed him up. Rather than admit his own disgusting behaviour he would gaslight me everyday about being “genetically defective” and having social phobia to the point that I internalised it and actually became socially phobic and could barely speak to people. Even when patients write on Google reviews how nasty his behaviour is, some of his family members put in positive reviews to cover it up (though overall, to some extent, they admit his nature because of how public it has become). It pisses me off.
RB is manipulative as hell: he tells 5 different stories to 5 different people to protect himself: to some people he portrays me as a nasty, morally bankrupt kid (he’s full of bad habits himself). To others, he talks about how my mind has been “poisoned against him”. To others, how he’s “worried about me”. He utters this rubbish to his family members and others and then they come to my mother with stories like “RB cried so much for registeredforthissite. He loves him.” and try to goad me towards him. Imagine being butchered by a man and then being told he loves you and try to be goaded towards a psychopath. All that would result (and decades of experience has taught me this) is a continuation of that manipulative behaviour. RB is so manipulative and abusive, that you lose your psychological balance, become unhinged, aggressive and then that is used as proof of insanity. RB is a urologist. If he was a lawyer, he’d use law against me. Since he’s a doctor, psychiatric blackmail has become a convenient tool.
I couldn’t take any of this any longer. All the psychiatry crap from my past, the diagnoses, the SSRI induced manic episodes and the stuff that took place during that time were being held against me to blackmail me into silence.
I met Mr. SAD and mentioned all this and told him that it’s ridiculous to take a kid traumatised from an abusive upbringing, label him as “bipolar” due to drug induced effects, institutionalise him etc. instead of doing something like asking the mother to leave for the sake of the child first, do something about the perpetrator and help him get justice. I wasn’t speaking specifically about only him, but overall what the sort of “help” from his institution had been.
Mr. SAD was flabbergasted and felt “accused”. His “advice” then became even more perverse: “your father may be a nasty and crude man, but every boy can do something about it”, “at least you have a father, I didn’t have one”, “he screwed your life and now you want to screw his, but one day his affection will come”, “go kill yourself if you want to”, “I see thousands of people” (note: so does my dad, and he’s still what he is) etc. Not to mention he started character assassinating me (“ungrateful”, “arrogant”, “privileged”) and invalidated everything I was going through.
I felt completely helpless and I was totally backed into a corner. I ran around like a vagabond to different cities to meet mental health advocates and activists to beg for help and protection from my parents and psychiatrists. I had nothing left at this point.
Shortly after, I stopped seeing Mr. SAD. I bid him goodbye and good riddance because I had had enough. The only major thing that came to me from his profession was the complete and utter re-abuse of an already abused person.
No degree of deathbed affection from RB will bring back what I have lost in my life and I am so traumatised, volatile, damaged and my marital, academic, economic, social opportunities and life have all been perma-damaged. Now, I have turned into an aggressive freak who speaks in the air like he’s hallucinating (though I’m not) because I’m playing out the mental trauma from my past and potential future scenarios in my mind. It’s become a mechanism to keep sane. All the things I couldn’t say to my dad or might have to potentially say, whatever I want to scream, I involuntarily end up playing it out. I have to consciously control myself from doing this sometimes and I look like a freak to people. The thing that hurts me the most is that I was a studious, academically oriented kid who has now lost out on that very aspect, i.e. academics. RB used to insult me about this too: how he’s a great Urologist and I’m a nasty kid who doesn’t study when actually I’m barely able to concentrate from gaslighting and trauma. My mother is a supportive person overall (I wouldn’t be alive without her) but a chronic hoarder who starts muttering nasty insults every time her hoarding is brought into the picture. She has repeatedly scapegoated me to win fights between her and RB which resulted in me getting even more abused by him. She ends up bringing out a side of my personality I hate. I love her but maintain a relationship from a distance because it ends up in shouting matches. I live in isolation and work remotely doing what I can amdist the breakdowns I have. If I were with any psychiatry guys, I’d probably be ECTed into oblivion by now apart from being labelled with god knows what else. What will I do with deathbed affection? I’m in my 30s, RB and Mr. SAD are retired in their 60s and the latter might even be dead soon given the hearsay that he’s already had cancer once (no idea if it’s true).
I’m cursed to have escaped an abusive psychopathic Urologist father to have ended up in the hands of a heartbroken fatherless MRPsych like some “buy one, get one free offer”. Rather than get justice I ended up being gifted double damage. I couldn’t see it earlier but what Mr. SAD engaged was in was also simply a form of gaslighting (inadvertent intitially, unlike RB) and transference. I’ve spent half my life first running from RB, then running from Psychiatry.
What kind of garbage is this from medical doctors? Too much power to get away with almost anything.
It is hypocrisy of psychiatrists to be a part of institutions like Cochrane and primarily focus on systematic reviews, meta-analyses etc. without doing anything to practically solve the day-to-day problems their clients face. They never talk about how badly their psychiatric categorisations affect people, how psychiatry is used as a weapon of blackmail, how much institutionalisation in some cases can traumatise a person, how psychiatry can make it nearly impossible for a person to get justice etc. Instead they talk about big bad antipsychiatry, Scientology, Tom Cruise and rubbish like that. They turn it around on damaged clients and claim they’re keeping suffering people from getting the help they need. Rubbish. Patients are sometimes even worse than the psychiatry “doctors”. A person with a relatively milder, less stigmatising “diagnosis” will rant and rave about how great his psychiatrist is and invalidate people who speak about how damaging it is for others in a situation different than theirs.
I remember asking Mr. SAD questions like: “Sir, why do you think there are people who talk about the damage of psychiatry, that their stories are similar regarding the problems they’ve faced and there are people who call themselves survivors of psychiatry”? Now, even I agree that “psychiatric survivors” is a bit of an excessively victim-ish name, but their experiences are not always untrue (even if people sometimes lie and exaggerate). However, his answers used to be like: “well, if you look, there will also be people calling themselves healthcare survivors”. Seriously? Firstly, I’ve barely seen people call themselves that. Sure, there are people who have botched surgeries and stuff, but that’s human error. Healthcare and psychiatry are not even close to being the same. I did not know kidneys can get personality disorders, livers can get mood disorders, lungs can get anxiety disorders and hearts can be oppositionally defiant disordered. I don’t see people telling other people they dislike “Hey! You have cystic fibrosis!”. No, they say “you’re insane, schizophrenic, borderline etc.”. Psychiatric diagnoses are simply used as slurs and psychiatric institutionalisation is used as a method of blackmail. You can’t do much of this with other medical branches.
Honesty and basic logic seem to be the first things to go in Psychiatry. It’s all about damage control because you know how terrible a lot of it is: doctors, their indoctrinated patients and families included.
Report comment
After the ex-Director of Cochrane South Asia and another interaction I had recently (I met another fatherless guy who told me “Even if I had an abusive father, at least I could have told him he’s a piece of shit”), I have developed a serious distaste for fatherless people who’ve lost their fathers at a young age.
Some of them are delusional and they think having abusive fathers who seriously damage their children (psychologically, socially, economically, emotionally etc.), sometimes for life, is better than having no father at all. They need to be provided some serious counselling, even some rough talk to take them out of their heartbreak fuelled delusions, especially before being made to sit as counsellors of any sort.
Is the ex-Director of Cochrane South Asia going to compensate me for the losses I have faced? Of course not. He has just conveniently escaped.
Report comment
Sorry for veering off of the topic of this article because this is an article on whether Cochrane’s reviews of psychiatric drugs are unreliable. But I need to finish what I’ve started. This article may on appear on search engine queries, so I’ll write it here.
If my experiences have taught me anything, I’d like to give some advice to people from what I’ve learnt so that they don’t end up in a situation like mine in their lives. This pertains to everyone, but more so to people from my country (India) and possibly to other similar developing countries.
i.) When you want to marry someone, don’t simply look to do so on the basis of their degrees or (potential) money making capacity. The fact that a person did a masters in anaesthesiology from X top rated institute or a Ph.D in aeronautical engineering from Y top rated institute may be less relevant than you think (even if X and Y are the topmost government/private colleges in the country with the hardest of entrance exams). First of all, education does not start at the level of engineering, medicine, law etc college. By the time you go to college for even undergrad you’re already 17 (or more). That’s 17 (or more) years of education prior.
You must see the totality of where a person is from: what kind of environment/society they were raised in since birth, what kind of school(s) they studied in, what their family background is, how many siblings they have, what their relationship with their own parents and siblings are like, what their social and religious values are, what kind of friends group and colleagues they have, what sort of health or financial issues they have, what kind of media they consume, what kind of jobs they have done (and in what kind of places), their criminal background (even unrecorded) etc. Most importantly, their personal proclivities and character as an individual too. People have different personalities and are attracted to different things. Siblings who grow up in the “same family environment” can have very different preferences and be attracted towards different things in life. Education and knowledge come from all the various spheres previously mentioned and not just colleges. If any of the aforementioned factors bring up red flags, terminate the potential relationship immediately irrespective of the credentials of the person, even if it hurts their ego and they start resenting people like you. Your life is more important than their temporarily bruised ego.
Getting into many of the top-ranking institutes and performing technical jobs where the mind is taxed (over simply the body) requires functionality, concentration, alertness and an ability to handle stress/anxiety more than merely intelligence (the latter is also necessary in reasonable amounts of course): the ability to study and work long hours and to rapidly solve objective bits in entrance exams in quick succession in order to get into those institutes in the first place. It is not a guarantee of extreme intelligence and not a guarantee of wisdom or character at all (some people have those, others not so much). You see people from top rated institutes even with degrees in engineering, medicine, science, commerce, civil services, law etc., sometimes up to the Ph.D or equivalent level, who may also be skilled and successful in their specific domains, utter completely untrue/anti-scientific nonsense or have crackpot beliefs and awful behaviour. In poorer/underdeveloped countries, such academic success (which then brings financial and social success) automatically brings a lot of fandom and their degrees are used as weapons to justify even untrue nonsense and nasty behaviour: the notion that because someone has A or B degree from X or Y highly ranked college or is very professionally/financially successful, it automatically makes them right. This also emboldens other horrible people because they rally around the successful person. A person could be very skilled at perfoming a kidney transplant, flying an Airbus A320, writing computer programs or designing rocket engines, but be extraordinarily ignorant in other ways. This applies to everyone (me included of course).
ii.) After getting married, do not have kids immediately. Live with your partner and observe their behaviour for at least 2 years first. Everyone is nice in public and everyone puts on a façade initially. But you only really get to know a person properly when you live with them for extended periods of time. Only then can you know what habits they have, how they respond to situations, any secrets they may be potentially hiding, how they manage their money, their values and beliefs etc. You don’t want to end up in a relationship with someone horrible and then have kids with them. It becomes abject suffering for everyone involved. Only when you are satisfied with your partner’s beliefs/values/habits/behaviour should you think of starting a family. If need be, it’s much easier to cut a relationship off when there are no kids involved.
Report comment