People diagnosed with severe depression show the same changes in brain scans when they respond to a placebo as they do when they take an actual antidepressant, according to a new study. Researchers also found that those whose symptoms were decreased by a placebo were more likely to report relief from antidepressant drugs.
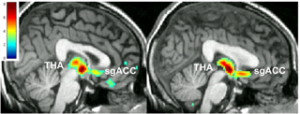
Right: PET Brain scan of patient response to antidepressant
“A possible interpretation of this finding is that such correlation is accounted for by the fact that, nested within the response to active antidepressant therapy, there is a substantial placebo response,” Dr. Maurizio Fava of the Massachusetts General Research Institute wrote in an accompanying editorial.
The researchers, led by Dr. Jon-Kar Zubieta, observed 35 patients diagnosed with untreated major depression through two placebo phases and active treatment with antidepressants.
In this single-blind study, the participants were initially given a placebo pill for two weeks but were told that they were taking a substance that activated internal brain mechanisms that fight depression symptoms. When they came in for a brain scan- done by positron emission tomography (PET)- they were also given a harmless injection of water and told it had fast-acting antidepressant effects. Following the placebo part of the study, the participants were given an antidepressant and were evaluated every two weeks over a ten week period.
The researchers found that participants who responded to placebo, activating their “mu-opioid system” (the brain’s natural pain killer response), were most likely to have a similar response when given an active antidepressant.
“The fact that placebo-induced endogenous opioid release in the network of regions mentioned here was associated with better open-label antidepressant treatment response may be simply accounted for by the fact that the placebo response in the lead-in phase was the same type of response observed following open-label antidepressant therapy,” Dr. Marta Pecina, the study’s lead author, wrote.
If a substantial portion of the neurological response to antidepressants can be accounted for by the placebo effect, this has serious implications for randomized control trial studies of antidepressant effectiveness. The results of this may already be apparent in a recent reanalysis of antidepressant efficacy versus placebo in major depression, which found that previous estimates of antidepressant efficacy compared to placebo had been significantly overestimated.
In this new study, the placebo effect predicted nearly half of the variation between individuals who responded to antidepressants after ten weeks.
“These results suggest that some people are more responsive to the intention to treat their depression, and may do better if psychotherapies or cognitive therapies that enhance the clinician-patient relationship are incorporated into their care as well as antidepressant medications,” Zubieta told the University of Michigan.
“We need to find out how to enhance the natural resiliency that some people appear to have.”
*
Peciña, M., Bohnert, A. S., Sikora, M., Avery, E. T., Langenecker, S. A., Mickey, B. J., & Zubieta, J. K. (2015). Association Between Placebo-Activated Neural Systems and Antidepressant Responses: Neurochemistry of Placebo Effects in Major Depression. JAMA psychiatry, 1-8. (Full Text)
Fava, M. (2015). Implications of a Biosignature Study of the Placebo Response in Major Depressive Disorder. JAMA psychiatry, 1-2. (Full Text)














To refer to SSRIs as essentially no better than placebo is misleading because while it’s technically true in clinical trials or in the ways they’re referring to in such analysis as the above article — SSRIs are also very different than placebos…they are NOT inert…they are very active and they make EVERYONE feel differently (one way or another).
People who are very depressed like to feel differently. Different can sometimes feel better even if only for a while..those folks who like it turn that difference into a “positive” placebo. Sometimes different feels much better and sometimes different feels really bad and even dangerous (think homicidal and suicidal–SSRIs are associated with both).
The fact remains SSRIs are not inert and they will act in all manner of ways in different people. And, then of course, yeah, they also cause iatrogenic damage…often severe…placebos don’t cause long term disability and brain injury.
The placebo meme is therefore overused and extremely misleading….it makes it sound like SSRIs do nothing at all which is absolutely not true. They are potent psychoactive and neurotoxic drugs. They can sometimes make people feel better and sometimes they make people violent, suicidal and homicidal. That’s quite a feat for something that’s referred to as a placebo, eh?
Report comment
I absolutely agree. These studies that say antidepressants are equal to placebo are dangerously misleading. This makes people not see the harm in simply trying one of these brain damaging drugs. I’m not sure I understand how this study came to this conclusion.
A study has already confirmed that just a SINGLE DOSE of an SSRI dramatically changes a healthy person’s brain structure. Connectivity was reduced in almost every area of the brain from the dose of SSRI.
http://www.newsweek.com/antidepressants-can-change-your-brain-immediately-study-271371
The most interesting thing about the single dose study is that it was done on non-depressed people who had never taken an antidepressant before. Therefore it showed how dramatic the changes begin from healthy to possibly permanently abnormal (damaged).
Brain studies on depression and other mental illnesses are some of the most misleading studies out there. Most of the studies are done on people who are on or had been on brain altering psych drugs for years or decades even. Then when researchers find brain abnormalities they blame it on the diseases instead of the real cause which is the drugs.
Report comment
I think the conclusion of the article was not that there is no harm in taking SSRI medication. I believe it was to show that a patient may have better results if they participated in CBT and built an ongoing relationship with a therapist than if they simply used drugs to treat their depression. The great part of the drugs effectiveness seems to come from the patients belief that it will work. If the patient believed that CBT would work then they would get similar results as if they took the drugs, perhaps better results because they would be working through the causes of the depression and simply medicating the symptoms.
Report comment
And also because of the lack of adverse effects, which are very real and can be extremely damaging. It’s one thing to take a placebo like Niacin that just gives you a facial flush and some discomfort. It’s quite another to take a “placebo” that can result in agitation, violence, anorexia, or permanent loss of libido, among other fun things.
—- Steve
Report comment
What a waste of time and resources antidepressant research is.
Time and money could be put into early childhood education, improving our infrastructure, helping homeless people, providing peer support and psychotherapy, making college more affordable, researching renewable energy….
But no, it has to be spent on fake medications to treat fake illnesses. The opportunity cost is staggering not to mention the harm done to real people needing real interpersonal help.
At least this study is showing how relatively worthless antidepressants are for most people, so it has more value than the big pharma trials.
As for this quote, “A possible interpretation of this finding is that such correlation is accounted for by the fact that, nested within the response to active antidepressant therapy, there is a substantial placebo response,”
What a novel idea! DUH… most people (except some psychiatrists) know that most of the supposed response to antidepressants is a placebo effect. Kirsch’s and Moncrieff’s research is pretty clear. I love how psychiatrists say these things like they’re groundbreaking or new.
It’s a good thing I don’t attend psychiatry research conferences.
Report comment
BPD, for all your talk about “studies” being the only real proof……
Report comment
How much can they really tell in those brain scans? I was told over and over I had a “normal brain.” They didn’t see any damage, and whatever was found after ECT I’m sure they wouldn’t have admitted to me. One neuro saw seizures. I got diagnosed with temporal lobe epilepsy, which was later discredited because they told me the seizures were a minor temporary drug effect. Sadly, I spent years with a shrink who went on and on about “the personality of someone with TLE….” Crap. Just stay away from those diagnosers.
Report comment
“How much can they really tell in those brain scans?”
Not much really. There are serious methodological issues with imaging, especially functional imaging related to artifacts and bad statistics (I always refer people to the IgNobel on the thought processes of a dead salmon – google it ;P).
But even when the study’s done rights all that imaging observes is a sum of activity in broadly defined brain regions. It tells you very little of what is actually going on and does account poorly for individual variability which is huge. We know that for most people for instance fear responses are controlled by amygdala but that is a huge oversimplification. There are different circuits within that structure, some activating, some inhibitory and that does not even account for the fact that it may be largely different for different people. Seeing that giving someone a drug makes amygdala more active may give you a hint this person’s more fearful but the easier and more reliable way to get that information is to ask.
In other words giving people drugs or placebos has some influence on their brain activity. That’s hardly an earth-shattering discovery.
Report comment
Those brain scans studies are ALL correlational. They take averages over time with a lot of subject and then highlight correlational trends. Not everyone with the “disorder” has the brain activity they show you, nor does everyone who shows this brain use pattern have symptoms of the “disorder” in question. There is no attempt made to determine causality (did the person’s brain become this way because s/he was feeling depressed or thinking about depressing things, or did the brain becoming like this cause the depressed thoughts or feelings.) And, as B points out, they are only usage patterns and reflect nothing at all about structure or chemistry underlying the pattern.
Other than that, they’re super reliable.
—- Steve
Report comment
I studied the life of two men who were known for their huge appetites. These were both such oddities at the time that all kinds of mythologies were passed around regarding their voracious eating habits. One was a Frenchman the the other I cannot recall. These men were both studied extensively. One was imprisoned at one point and was said to have eating live rats and cats, but of course that could have been a rumor. The reason the lives of these two men interested me is that I myself have suffered from horrific binge eating (quantity-wise, that physically disabled me to such an extent that I was okay with going on disability. I also from pica, for which I was deeply ashamed. This happened to me as an adult, not a toddler, and this wasn’t related to a pregnancy. So I was curious as to what conclusions had been drawn regarding these men who must truly have suffered (I should know!). As it turned out, they were saying one may have had a TBI to the amygdala. I have also learned that there is no “appetite center” to the brain as originally thought. There is indeed an appetite center but it isn’t at one specific place. Meaning they just don’t know. If the folktales about them were true it does indeed sound like TBI had caused it, given the extremes to which they ate.
Meanwhile, I have studied binge eating extensively, since I was determined to solve it. Of course, mental health did nothing to help me whatsoever, but taught me that I had to follow their orders and that they knew what was best for me (baloney!). The only reliable way to do this was through extensive self-experimentation. I could only do this far, far away from psych and their diagnoses. I concluded that no matter what weight a person is at, most likely the behavior is a result of malnutrition. Most often, crash dieting precedes binge eating, and the behavior is a result of the severe malnutrition that has developed from dieting. Another thing that is never considered is that some people have increased need beyond the norm for certain nutrients. This is often hereditary. In the care of eating disorders it’s so rare to question a patient about food preference, and focus on the negative behaviors only. Rarely do the professionals take into account family history and cultural or religious significance of various foods. They’ll only ask if anyone has ever committed suicide or had “mental illness” or alcoholism. I’ve even seen them give foods the patient is allergic to that are ingredients in “tube feed,” or insert a latex tube into a patient who is known to have latex allergy. They almost always completely miss the boat. I do not see “eating disorder” as a “mental illness” but a nutritional difference. Any “mental illness” is learned in care or secondary to bad eating habits. Conversely, it’s possible to develop terrible eating habits from being in ED “care.” I have known several folks who were pushed into this “care” when in fact no one was listening to their logical explanation for unusual thinness. It’s completely ass-backwards. I am determined to change how things are done. As long-term sufferer I do have expertise because I have lived it.
Report comment
Maybe because it is placebo effect for the most part?
That being said I don’t really buy the brain imaging studies at this point, not until they get reproduced independently multiple times and preferably with different, complementary methods.
“Anti-depressants” likely have more effects on the brain than placebo, because they screw up the neurotransmitter signalling.
Report comment