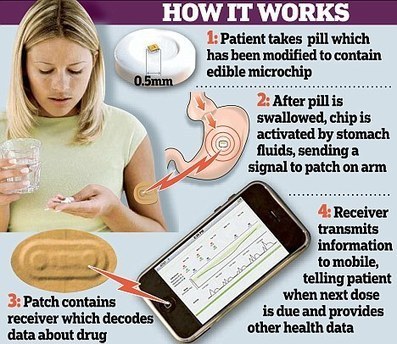
The FDA has rejected the drug/device combination designed to monitor patient adherence with Abilify from Otsuka Pharmaceutical and Proteus Digital Health. Just last week the FDA issued an additional warning that Abilify (aripiprazole), an atypical antipsychotic, could lead to compulsive and dangerous behaviors.
-
Scary to read this type of language:
“The idea is that patients with mental illness, whose day-to-day functioning depends upon adherence to their medication, are a good initial target and proving ground for the Proteus technology…”
Are psychiatrists not ashamed to be affiliated with companies that act in this way?
-
A temporary victory. Sad in a way, this was such a great propaganda tool for us. The Onion couldn’t have done better.
-
Oldhead: “Sad in a way, this was such a great propaganda tool for us.” That is very callous, triggering, insensitive thing to say, considering some of the readers on this site are involuntarily treated and forcibly medicated! Maybe you better get up from your armchair one day and do some field work: i.e. help out at your local soup kitchen, help someone in crisis on a hotline, etc. You might be reminded of what is at stake. The people who are at risk of being given these creepy medications are real people, not pawns in some propaganda war. I for one, think your comment should be deleted as innappropriate!
-
Sorry you don’t appreciate my irony. I’ve been amply forced-drugged in my time, thank you, and thus consider myself qualified to engage in “mental patient” humor. Besides, it was and is still a great propaganda tool, nothing wrong with that. It’s about time they gave us something.
-
-
-
I still think that just dropping one of these creepy Abilify chip pills into a cup filled with warm grapefruit juice or vinegar would simulate the stomach acid needed for the sender chip to tell the pharma gestapo the pill was ingested.
Look at all the web pages for beating drug tests, that tells me that information on how to beat chip pills , methods and devices to do so would hit the internet soon after these pills came out anyway.
-
Quite likely. I do think I remember something about it also reporting your body position at the time of ingestion.
-