This week we talk with Professor Jim van Os and Doctor Peter Groot about their latest study which looks at the effectiveness of tapering strips to help people get off antidepressant drugs.
Jim van Os is Professor of Psychiatric Epidemiology and Public Mental Health at Utrecht University Medical Centre, the Netherlands and Peter Groot works with the User Research Centre of UMC Utrecht.
They are both involved with the development and study of tapering strips which are pre-packaged, gradually reducing dosage tablets that facilitate tapered withdrawal from psychiatric drugs. In this interview, we discuss their latest research which examines tapering strips in real-world use.
The transcript below has been edited for length and clarity. Listen to the audio of the interview here.
James Moore: Jim and Peter, welcome. Thank you so much for joining me again for the Mad in America podcast. We are here to talk about your recent paper entitled “Successful Use of Tapering Strips for Hyperbolic Reduction of Antidepressant Dose: A Cohort Study”. This is published in the journal Therapeutic Advances in Psychopharmacology.
I believe this is the third study to look at the effectiveness of tapering strips and importantly, in real-world use. Peter, could you sum up the study for us? How many participated, how was the trial undertaken, and what were the results from it?
Peter Groot: I’m happy to do so, but I’ll start by explaining how we did the study. Our study was a retrospective cohort study and we could do the study because, in the Netherlands starting in 2013 when the first tapering strips became available, a growing group of patients in the Netherlands had used tapering strips because the doctors prescribed them to taper antidepressants.
This gave us the unique opportunity to ask them to fill in a short questionnaire after they had finished the tapering strips they had been prescribed. To get a high level of response, we kept the questionnaire deliberately as short as possible. One important question we asked was ‘did you experience withdrawal symptoms during the taper?’ The answer could be a number, from one to seven, where one is ‘not at all‘ and seven is ‘very much‘. These answers gave us the indication of withdrawal during tapering with the use of tapering strips.
The second important question we asked was: ‘How did tapering go?‘ Again, the answer was a number from one to seven, where one is ‘very well‘ and seven is ‘very bad‘. This answer gave us an indication of how patients perceived the tapering process when they used the tapering strips.
This was not all, because, in our large sample, more than 60% of all participants had tried to taper their antidepressants in the past without the use of tapering strips. This allowed us to ask all these people the same two questions again, but this time, about the previous tapering effects.
This resulted in a large cohort of patients who could compare tapers without and with the use of tapering strips, and this gives a so-called ‘within patient comparison’, which made it possible to directly compare tapering without and with the use of tapering strips.
Very important is that the results we got were the results from the patients who were being treated in daily clinical practice.
Jim van Os: So then, people ask the question ‘how big was the effect size?’, that’s what people want to know.
Basically, to give you an idea, if you look at the distribution of withdrawal when people use tapering strips versus when they don’t, you see the complete opposite shape of a distribution. So with the tapering strips, it is mostly in the area of ‘went very well and was okay’, and without tapering strips, it was in the area of ‘really poorly and lots of withdrawal’, et cetera.
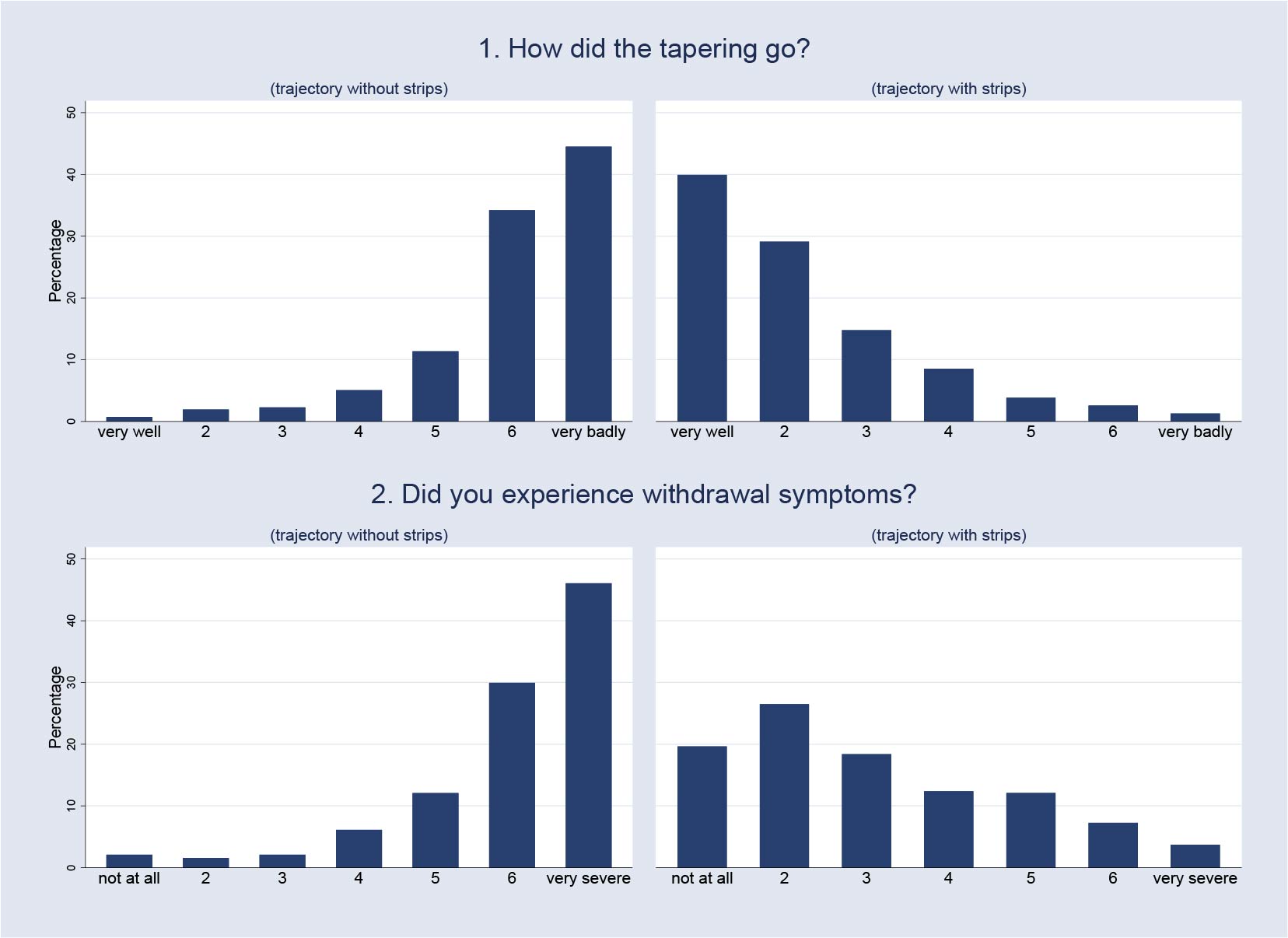
So, a very convincing effect size. And then people will say: “This is not a representative sample” which of course is true. These are the individuals who have tried previously one or two or three times and failed and then had to resort to tapering strips to try to stop and succeeded. Then, of course, you have a selection of the people most in need of tapering strips.
So our reasoning is, if it works in that population who have the most need for care with regard to withdrawal, then it’s very likely to be also successful in people who have less intense needs regarding withdrawal.
Moore: I was interested to see that, across the three studies, the results are remarkably consistent, around the 70% mark, is that right?
van Os: That’s correct James. We were actually surprised to see how consistent the effect size was, particularly when we compared study one with study three, which had completely identical methodology and questionnaires but different people across different time periods.
So, this is one way to think of science. Some people say, “You should have a randomized controlled trial”. But a much stronger way of providing evidence is replication, which we all know. Replication across different samples, different timeframes, different GPs, different patients, et cetera.
Moore: I read that the study data was anonymous. So, people didn’t have to identify themselves when they responded as to the success or not of their withdrawal experience.
Groot: That’s correct, yes, we know something about it, because they have been prescribed tapering strips, but the setup was such that they gave their data anonymously.
Moore: In reading through the paper, I was interested to see that you collected data on the amount of time the people had been on their antidepressants.
The median usage time of your sample was five to 10 years. That seemed to me to go against some of the prevailing wisdom that long-term users might always need to take many months or years to come off because I think the median time taken was 56 days or two tapering strips. I know the longer-term users tend to take more time to come off, but even counting that, there was quite a proportion who were successful. Is that right?
Groot: That’s right and you can say that in general, based on our data, the number of years people have used the antidepressants, has predictive value for the amount of withdrawal symptoms patients can suffer when they taper.
It is very important not to over-interpret these findings. This is because patients differ very much from each other. You’re dealing with a very heterogeneous population. A population in which perhaps a large number of patients seem to be able to taper rather quickly without having many problems. A smaller group will experience more problems and a still smaller group will experience very severe problems.
Now the problem here is that we do not really know how large these percentages precisely are. Because this has never, until recently, been investigated properly. Only in the last couple of years there have been studies which have investigated this in a much better way.
The conclusion that I draw from all these data is that withdrawal problems have been greatly underestimated as they have been reported in the literature. And that all the figures, therefore, are probably too low.
I don’t think that it is really a big problem that we do not know precisely how severe withdrawal problems are and how many patients suffer from them. This is because, even without this knowledge, it should be possible to help patients who wish to taper responsibly. Because it is much more important to be able to follow patients during tapering and to be able to adapt the tapering schedule when the patient starts experiencing problems during the tapering.
Moore: In addition to looking at the length of time that people had been on the drugs, you also recorded previous failed taper attempts, which seems to be another important factor. It was clear that a larger number of previously failed attempts was linked with a lower success rate in getting off.
That strengthens the argument to provide the best possible tapering method as early as possible in people’s withdrawal experience so that we minimize those failed attempts because it makes the hurdle greater to get over later, doesn’t it?
Were there any other factors that were linked with a reduced success rate for people trying to get off antidepressants?
van Os: So the length of use and previous withdrawal really stand out as the two factors contributing most. And there is a suggestion, which I think is interesting in two of the studies, that paroxetine may actually be harder to come off versus any other antidepressant. This is something we saw more conclusively in the last study, but there was also a suggestion in the first study. So this is something that we want to keep track of.
What really stands out more strongly is the longer the previous use of antidepressants, the lower the success rates. That’s the most important factor.
Moore: I noticed that there was quite a high proportion of females in the study. Is that because more females are prescribed to in the Netherlands so the sample would be bigger because of that, or is it because females generally find it more difficult to come off? Have you any insights into why that might be?
van Os: Actually, what we did in the third study is that we compared the demographics of our sample to the demographics of a very large random population sample that has been collected in the Netherlands recently, and that also had items on antidepressant use. We found that the demographics of our sample in terms of age and sex were similar to the demographics of the antidepressant users in the general population sample.
However, the variables that were skewed in our sample were the type of antidepressants, and this will not surprise you, the rates of venlafaxine and paroxetine were higher in our sample compared to the general population sample, which is why we have been telling general practitioners, ‘please do not prescribe venlafaxine or paroxetine anymore‘. There is no need.
Moore: Have you had any response to that request?
van Os: Actually, people are really surprised. The general practitioners I speak to still have a different image when they think of venlafaxine or paroxetine as very effective antidepressants, which is of course because they were marketed very proficiently and professionally in the Netherlands. I think this was very successful in the sense that this created an aura of them being very successful, and this has persisted until this time.
Moore: We know that as it currently stands, people who insist that they want to taper slowly will probably be prescribed a liquid form. Are there any studies that you are aware of that have looked at the effectiveness of liquid forms for tapering use?
Groot: Regarding the use of liquid medication, I think it is important to make a few remarks first.
My first remark is that it is possible to taper drugs using liquid medication because we see that there are patients who succeed when they do this. But there are also patients who try to taper using liquid medication who don’t succeed, and who have big problems when they try. But we don’t know how many people succeed and how many people fail. We just have opinions of doctors about this and we hear stories from patients. I hear stories from patients, and stories I hear that apply to a number of patients who are having trouble using liquid medication, especially when they are nearing the end of the taper.
My second remark is that, as far as I know, we have no reliable data about this. Because this has simply never been investigated properly.
My third remark is that liquid medications have not been developed for tapering and they have never been tested for this purpose. I asked this to GlaxoSmithKline about paroxetine and they told me: “Actually, we don’t know, liquid paroxetine was registered 20 years ago. Below the lowest registered dose, we cannot guarantee that liquid medication will work properly.” So basically, they did not know.
I do fully understand why patients ask for liquid medication. That is because the medication dosages they want are simply not available. So, it’s a matter of having something better than not having something at all. It is the only viable alternative that patients and doctors can come up with currently, and that this is so is because pharmaceutical companies have not given them anything better over all these years.
My next remark is that although doctors and patients may think that liquid medication may help most patients, nobody knows for how many patients this is true and for how many patients this is problematic. It may be problematic because it may be difficult to follow instructions adequately, which may be especially difficult for more vulnerable patients, or for patients using a number of different medications at the same time.
Taken together, this gives me very mixed feelings about the use of liquid medications. And I wonder what patients would prefer if they would be given the choice between liquid medication or the use of tapering strips which are much easier to work with, much easier to understand. Easier for patients who have trouble with using liquid medication properly and also because the dosing is much more accurate.
Moore: Turning to this whole issue of comparing withdrawal methods, I’ve seen discussions on social media that suggest that, until a randomized controlled trial is done that compares all the different ways that people can withdraw, then we won’t be able to tell if there is a standout method or not.
I just wondered what your feelings were about it. Is a randomized controlled trial the best instrument to use to assess the wide variety of experiences that people have when they are trying to stop antidepressants?
van Os: I think this is very interesting because this is really a patient issue and then about how does the scientific community react to that. How do they think they are going to help patients to answer this question. There are different ways you can think of a trial, of course, but the trial they have proposed in the Netherlands, that may take five to 10 years and likely will be inconclusive, is that they want to compare what they call ‘regular tapering’ of venlafaxine and paroxetine which basically means cold turkey from the lowest available registered dose because there are no smaller dosages to taper.
So this is what they call ‘regular tapering’, just stopping your medication at the lowest available dose for venlafaxine and paroxetine and then see what happens, and then compare that with personalized tapering.
The problem is that they will likely run into trouble with that design. This week, there was an article in JAMA, and this was about opiates, and they were actually describing the fact that it is impossible, probably, to do a randomized tapering trial. Why? Because of ethical issues, because the outcome of the trial is not that you bring something good, like trying to cure your cancer, but to avoid something bad. So then if the control condition is that you let the bad thing happen and then you see if less of that bad thing happens if you do something less bad, that is completely unethical.
And the interesting thing is that the Americans, of course, are much keener on these ethics issues because ethics is a much more litigated issue there. Whereas in the Netherlands, and I’ve also seen people writing about this from the UK, the approach is ‘let’s see who gets the most withdrawal if you do this or that‘. This is simply not possible, ethically.
So the only reasonable randomized controlled trial one could possibly undertake is to compare personalized tapering with a tapering strip and compare that in a randomized controlled trial with a personalized form of liquid tapering. So, the two arms are both personalized, but one with liquid form and one with tapering strips. That would be a reasonable trial. The only thing is that the ethics committee might say ‘will you be able to show that these liquid forms can be dosed precisely?’, and the answer is we can’t, because that’s why the consensus document on tapering in the Netherlands actually does not allow for liquid tapering. They disrecommend it because they say it is too messy and you can’t guarantee that the small doses in these droplets are actually the ones you’re actually supposed to take.
So, ethically, I think that would be difficult as well, and then the third argument is simply, scientifically: it’s been known for a long time that proper observational studies and trials yield the same results as randomized controlled trials and the advantage of observational studies is that, of course, that the populations are much more representative than in randomized controlled trials.
There are many more problems I could go on about. I don’t think a trial is going to be conducted like the one they proposed here in the Netherlands. It’s simply not possible, ethically.
Groot: We think that tapering should be personalized and that we should try to avoid a one size fits all recommendation and guidelines, but this is what is currently sort of happening. But of course, what works for one patient is not automatically going to work for another patient.
As I said earlier, there is currently no proper way for a prescriber to predict this reliably. Therefore, it’s very important to self-monitor, tapering and act on the basis of this during the tapering. To make the tapering go as well as possible.
What I just said is not different from what patients have been advocating for many, many years now. I can specifically name Ed White, John Read and Sherry Julo who described Facebook groups that are helping patients come off psychiatric medications and Adele Framer, founder of Surviving Antidepressants, also wrote a very good article about this. Both of these articles were published last year and in my opinion should be obligatory reading for physicians who must help patients to taper safely and responsibly.
What I described I think is also basically what somebody like Professor Heather Ashton has been advocating for coming off benzodiazepines for many years. Her message was ‘do not simply follow a standard guideline but instead work together with the patient to guide the process of tapering. Take the time for it and listen to the patient and don’t try to go too fast.’
It is interesting to point out that RCTs are essentially a form of group research. So, if you do group research, the results you get will be valid for the groups you studied, this is something different than being valid for the individual patient within a group.
Moore: This says to me that randomized controlled trials are probably right where you’re asking a very specific question about a very specific population of patients, and yet that isn’t the world of withdrawal, is it? We’re talking about a variety of experiences and wide variation in how people might assess success. You can’t placebo control this because it’s unethical to let people think they are being withdrawn when you’re actually giving them the placebo, which is having no effect at all and therefore potentially harmful.
So, I think the studies that you are doing are probably the best method of assessing the effectiveness of tapering interventions.
van Os: Correct.
Moore: Looking to the future, I’m aware that there continue to be barriers to the adoption of tapering strips. Partly coming from perhaps the insistence that antidepressants are not dependence-forming drugs, but also partly from local healthcare arrangements. I just wondered if you could both share a little bit about what your experiences have been in trying to encourage the adoption of tapering strips in the Netherlands?
van Os: Yes. I think this is a very interesting topic because it’s basically like a PhD on medical sociology. In the sense that what happens in the Netherlands is that Peter, in 2013, proposed to 20 professors of psychiatry, the best-known professors of psychiatry in the Netherlands, saying ‘look, there is this problem, there is withdrawal, let’s write an article together and tell people there is tapering strips, and they can order them’.
Everybody was very enthusiastic. They were thinking this is great, this is a need. So we published the article together with Peter and then, what we saw is actually that people started using them. So there was a demand for them, and then with the demand, there came a signal and a debate about how large is this group.
So, there were fora and there were groups, and actually, it seemed that the prevalence was much higher than people had expected. With that signal came also a professional debate, in the sense that general practitioners who are responsible for 80% of antidepressant prescriptions were asking themselves, ‘did we do something wrong?‘ Then, psychiatrists were also weighing in, saying, “Is this really so severe?”
That professional debate had a certain kind of unease that was emerging. In the sense that admitting that withdrawal was actually prevalent, sometimes severe and requires a different prescribing practice was admitting ‘we haven’t been very careful with this molecule at all‘.
At the same time, what we saw is that there was a big debate about the effectiveness of antidepressants. So, lots of people referred then to the Lancet network meta-analysis, saying, “Look, it’s effective“, but if you look really carefully at that network meta-analysis, the real thing it’s saying is there is a non-clinical difference between placebo and antidepressants.
The reality is that probably some people have a really good response. But you can’t predict whom and a very large group will not have a response at all, but will have difficulties coming off antidepressants. So, the whole debate about withdrawal was also becoming a whole debate about psychiatry and the prescription of antidepressants.
Then, of course, we are Dutch, we don’t like spending money. We’re very mean. So when the group became larger and the demands became larger, the health insurers stopped reimbursing them because initially, they thought this is very nice, this is one or two people, and then we can all move on. But all of a sudden, they became aware it was really a societal issue. So, the financial components weighed in as well. It was professional, financial, and it was also about, I think, the underlying civil rights movement of patients, which in the Netherlands, we’ve always been very slow to acknowledge.
Groot: Once the tapering strips became available, I thought that they would be warmly welcomed, especially because all these professors supported us. And because it made possible what guidelines had always been asking, to let patients taper gradually when needed. But much to my surprise, the largest health insurers, who have about 90% of the market in the Netherlands, did not want to reimburse tapering strips.
This has led to a situation where all the responsible parties involved are pointing to each other instead of taking responsibility. This has lasted now in the Netherlands for more than five years, and there are court cases going on about it and we have no idea when it ends. At the same time, we see more and more doctors who are prescribing tapering strips.
So, it’s a very frustrating situation as far as I’m concerned.
Moore: I have to say that, as someone with lived experience, it was hugely disappointing in the UK to see that the biggest response to the need for tapering was the view that unless we know precisely the size of the population affected, then we can’t possibly do anything about it. There is a need, there is a solution, there is a way to help people, yet we can’t possibly help unless we know exactly the percentage affected.
Groot: This looks very much like the argument that was here. Basically, they are saying, if there are only a few people who suffer from it, then we don’t need the reimbursement. I compare this to a situation where one of 10,000 persons gets a specific form of cancer. Then we will not say this. Then we will say ‘there is a medicine for 100,000 Euros and it should be paid for‘ because this is an unmet need of this patient. But when it comes to tapering, we say, ‘no, it’s only a problem if a sizeable proportion of 20% of patients is suffering from it’.
It is as if we are saying that if the group of patients that has the problem is too small, they will not be helped. That is something I still can’t understand.
Moore: So we’re in a position where you’ve now done three studies amongst some 2,000-odd people. You’ve had a confirmed success rate of around 70% each time. You’ve collected very important and valuable data around the quality of experience and the length of time on the drugs. So, it seems to me that you’ve replicated your success rate on several occasions now.
What more do you think needs to be done, if anything, for this approach to be taken seriously by prescribers?
van Os: I think that what has helped greatly is that, for example, the media showing real images of real people struggling and showing the real suffering there, and I think this has helped enormously in our case for the wider population to understand this because people don’t know about it. They’ve never been told about it. So, if there is general population knowledge about this, I think that this will impact also on prescribers. Because then patients coming to the GP will say, “I don’t want paroxetine because it’s associated with severe withdrawal“. Or, patients will ask for tapering strips, and then the GP will ask, “What do you mean? What is a tapering strip?” Then he will look it up, et cetera.
So, I think with more awareness and use, the problem will resolve itself, but we have to give it another five years, I think, unfortunately.
Moore: Peter, your thoughts on the future?
Groot: I hope it’ll go a little bit faster. You have to get yourself together and say, “We’ll go on“, and this is of course what we do, with the studies we’ve done, but also with the studies that are coming. We will go on asking patients who have tapered sometime ago how they are doing now. Are they still off their medication? Or have they started using it again? The result of our second study, in which we asked this question, is that about 70% of people who have tapered an antidepressant completely one to five years ago (we could not measure longer) are not using the medication again. There seems to be no difference between patients who have used the medication five years or one year, or 20 years.
This for me is a very promising finding. Because it suggests that also people who have been using antidepressants for a very long period of time, if they get the opportunity to taper carefully and may take a long time to do it, it seems to be possible for them to come off the drug and stay off the drug.
Moore: That’s really important, isn’t it, because in support groups, we so often hear of people who have been told to come off by the standard guidelines. So, 50% reduction every two weeks or whatever it is, then they get into terrible trouble, they go back to their prescriber and their prescriber’s response is to say to ‘go back on the antidepressant’.
That poor person has not only had a terrible withdrawal experience, they are also no further forward from where they were. They are back on the drug, they’ve got to face that hurdle next time. So the fact that you are looking at longer-term outcome data in terms of if you have a better trajectory, an easier trajectory off the drug, you are more likely to stay antidepressant-free in the long term. That’s hugely significant, isn’t it?
Groot: That’s what our study suggests and I think that’s also what many people have already been thinking.
van Os: So, I was just thinking that we are also greatly helped by voices like David Taylor and Mark Horowitz who actually have professional and lived experience combined, and then actually tell us about that.
Psychiatrists telling other psychiatrists, pharmacologists telling other pharmacologists it’s real, it really helps. Also, I think it helps that we have places for epistemic injustice like Mad in America, which is basically a platform for the civil rights movement, which is really helpful. I think, for you Peter, to amplify your voice and be heard, that is really great.
Moore: I can’t thank you both enough for your work. I am delighted to see the 70% figure replicated again for the third time and one wonders how many of these studies have to be done with such a success rate for people to wake up and take notice.
Groot: May I make one remark here? The 70% of people who succeed in tapering antidepressants completely may give the impression that the other 30% has failed.
This is not true, because a number of these patients actually end up being on a lower dosage. Other patients may find out that it may be wise perhaps for them to stay on the drug they are using.
Moore: So, tapering strips can help people find a minimum effective dose. Is that right?
Groot: For instance. But also you can see that the patients, even the patients who are in this 30% group, of whom you can say they failed to taper completely still say that they are much more satisfied being able to use the tapering strips than when they failed tapering without using the strips. I think this is also a very important finding because it suggests that patients have a better insight into what they are doing.
***
















The need for support for trying to stop a brain drug, a drug that the prescriber has no clue as to what it affects, points us to not prescribing in the first place.
No one, not even top neurologists know how these drugs affect the body/brain. No one knows what changes are permanent.
The whole “treatment” paradigm is quite random, just as is the DSM.
Report comment
HI,
MY NAME IS RACHEL COLLINS, AND I HAVE BEEN READING ABOUT THE METHOD OF STRIP TAPERING FROM ANTIDEPRESSANTS , IN MY CASE I AM ON 75MG OF VENLAFAXINE AT THE MOMENT AND AM TERRIFIED TO DROP DOWN TO 37.5 OF THIS DRUG,
I AM A LADY OF JUST GONE 70 YEARS OF AGE. I AM DETOXING (TRYING) FOR 8 YEARS.
I LIVE IN DUBLIN , IRELAND. I WAS GIVE EFFEXOR BY A DOCTOR IN ITALY WHEN I WAS DEEPLY DEPRESSED, AND I DID NOT KNOW I COULD NOT GET OFF THEM. I HAVE BEEN TAPERING FOR 8 YEARS FROM 300MG OF EFFEXOR TO 75MG AT PRESENT.
I HAD TO GO SEVERAL TIMES BY AMBULANCE TO THE A AND E DEPT. OF MY LOCAL HOSPITAL WHEN I GOT DOWN TO 37.5.
AS I WAS GETTING SUICIDE IDEATION. AND HAD TO GO BACK UP TO 75MG OF EFFEXOR.
THANK YOU FOR THE INFORMATION I READ ON THE INTERNET.
DO YOU THINK MY OWN DOCTOR IN DUBLIN CAN PRESCRIBE TAPERING STRIPS FOR ME.
RACHEL COLLINS
[email protected]. IS MY E-MAIL
MY MOBILE IS 0857277461
Report comment