A new study in The Lancet found that transcranial direct current stimulation (tDCS) was no better than a placebo (sham treatment) for depression. As a multi-center, sham-controlled, triple-blinded study, this was a well-designed trial with results that should be considered robust.
The research was conducted by a large team of German researchers led by Gerrit Burkhardt at Ludwig-Maximilians-Universität München, Munich, Germany.
According to the researchers:
“Active tDCS was not superior to sham stimulation during a 6-week period. Our trial does not support the efficacy of tDCS as an additional treatment to SSRIs in adults with MDD.”
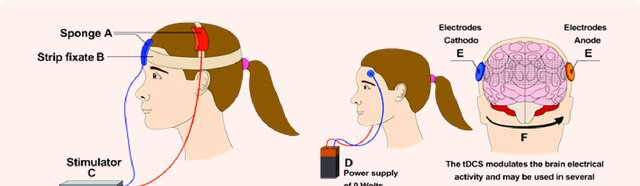
tDCS is one of several controversial brain stimulation or “neuromodulation” devices, which also include transcranial magnetic stimulation (TMS). TMS involves using a large machine to deliver electric pulses to the brain, while tDCS is a small device that delivers a constant, low electric current to the brain.
While TMS is FDA-approved for the treatment of depression and OCD, tDCS has not received FDA approval for any indications. tDCS devices are sometimes purchased online and used at home by people who call themselves “biohackers,” although websites generally include the caveat that users are not to personally use them “for medical purposes.”
The study included 150 people with, on average, moderate depression. All of the participants had “treatment-resistant” depression—meaning, in this study, that at least one SSRI had failed to improve their depression. In the study, they continued on the ineffective drug throughout treatment.
The intent of the trial was to see if tDCS, as an adjunct to ineffective SSRI treatment, would result in an improvement on depression rating scales.
Only about 7% of the participants had tried psychotherapy.
In the study, 77 participants received tDCS, while 73 participants received sham treatment, a fake version of tDCS designed to appear like the real thing. The participants received treatment sessions every day for four weeks and then two additional sessions per week for two more weeks (six weeks of treatment total).
The study was triple-blinded, meaning that none involved knew whether the participants were receiving real or sham tDCS. It was also conducted at eight different hospitals in Germany, which helps to give real-world validity to the results (since we know they are not driven by a single center’s experience).
There was no difference in improvement on any of the outcomes between the two groups at six weeks, including the primary outcome of the MADRS depression scale as well as the secondary outcomes of BDI-II, CGI-S, GAF, SF-36, SHAPS-D, and STAI scores. This remained true at both 18-week and 30-week follow-ups.
In terms of remission (no longer having depression), more people in the placebo group got better within six weeks: 38% versus 31% (this difference was not statistically significant). That is, about a third of those with moderate, “treatment-resistant” depression no longer have depression within six weeks, whether they receive treatment or not—and that’s despite having been continued on a treatment (SSRIs) that was already assessed as ineffective for them before starting the study.
Although tDCS was ineffective at treating depression, it did come with some harms, including—concerningly—an increase in self-harm behavior.
“The active group had higher incidences of headaches, sleep-related problems, local reactions at the treatment site (eg, burning sensation or skin irritation), restlessness, nausea, flash visions, and self-injurious behavior,” the researchers write.
Sixty percent of those receiving tDCS experienced adverse effects, while 43% of those in the sham group reported adverse effects.
Despite their null finding, the researchers write:
“Future research should focus on advancing the neurobiological understanding of the effects of tDCS on depressive symptoms and incorporating technological developments to establish individualized tDCS as an alternative to a so-called one-size-fits-all approach.”
This statement, a call for research on the supposed “effects of tDCS on depressive symptoms” and a call to “establish individualized tDCS,” despite finding that the treatment was no better than placebo, may be explained by the lengthy financial conflicts of interest held by many of the researchers involved. Some were funded by companies in the brain stimulation space, including Neuromod and Brainsway, as well as many other pharma and device companies. Some had been given unspecified brain stimulation equipment by these companies, which can run into the hundreds of thousands of dollars (although tDCS devices specifically are relatively inexpensive). At least one researcher on the study has a patent pending for brain stimulation devices and is a chair on the executive committee of the German Society for Brain Stimulation in Psychiatry.
****
Burkhardt, G., Kumpf, U., Crispin, A., Goerigk, S., Andre, E., Plewnia, C., . . . & Padberg, F. (2023). Transcranial direct current stimulation as an additional treatment to selective serotonin reuptake inhibitors in adults with major depressive disorder in Germany (DepressionDC): A triple-blind, randomised, sham-controlled, multicentre trial. Lancet. Published online July 03, 2023. https://doi.org/10.1016/S0140-6736(23)00640-2 (Link)














These devices are just scams. They do absolutely nothing, which is why you can freely buy them in the EU.
In that “study” a current of 0.2mA was used. That is nothing. Touching both poles of a 9V battery probably induces a higher current. One of those batteries could power such a device for 1000 hours, just touching both poles would probably discharge it faster.
Actual electroconvulsive torture uses a current around a thousand to tenthousand times stronger.
Report comment
Of course they are. Even if you’re a simple materialist you can find a multitude of things that can induce depression, because it isn’t a disease unto itself, like these shaky “treatments” advocates would have you believe.
Report comment
Psychiatry has a long history of being a factory of quackery. And things haven’t changed much.
Report comment
It is so refreshing to read this example of how an actual unbiased journalist covers a scientific article with the goal of uncovering the truth as opposed to generating clicks with sensationalist nonsense or pandering to paid sponsors. Thank you Peter for another outstanding article.
Report comment