The Reckoning in Psychiatry Over Protracted Antidepressant Withdrawal
Medically-induced harm—affecting tens of millions of people worldwide—has taken the field decades to take seriously.
How Academic Psychiatry Minimized SSRI Withdrawal
If academic psychiatry is evidence-based, why did it take two decades to recognize SSRI withdrawal as widespread and chronic among patients?
The P-Value Problem in Psychiatry
Stanford researcher writes that readers should check the effect size of results instead of looking at the p-value.
How Dissenting Voices are Silenced in Medicine
Researcher criticizes the many ways opposing viewpoints and dissenting voices are squashed in the field of medicine.
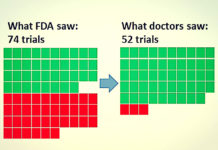
Where Are the Results of These Five Clinical Trials of Antidepressant Drugs?
The results of five large-scale clinical trials of antidepressants have never been made accessible to the public, a data set compiled by an international team of researchers shows. Their discovery highlights the incompleteness of available data on the safety and efficacy of antidepressant drugs.
Researchers: Antidepressant Withdrawal, Not “Discontinuation Syndrome”
Researchers suggest that the pharmaceutical industry had a vested interest in using the term “discontinuation” in order to hide the severity of physical dependence and withdrawal reactions many people experience from antidepressants.
Amsterdam Files New Study 352 Whistleblower Complaint
Jay Amsterdam, who first blew the whistle on corrupt research practices in a study conducted by GlaxoSmithKline (GSK) eight years ago, has now submitted...
Biogen Pushes FDA to Approve Failed Alzheimer’s Drug
A new analysis, published in Lancet Neurology, demonstrates how Biogen is spinning results from two failed trials for a new Alzheimer's drug.
Researchers Set the Record Straight on Controversial Zoloft Study
An issue of Lancet Psychiatry is devoted to clarifying the lack of efficacy for Zoloft (sertraline).
Medical Ghostwriting: When an “Author” Is Not Necessarily an “Author”
Ghostwriting, which is prominent in the psychiatry literature, is a scam in which pharmaceutical companies use an academic sleight of hand to stump the naïve reader. It is time for editors of the major medical journals to use the same standards of authorship found in the humanities and social sciences.
What is the Evidence for Empirically Supported Treatments in Psychology?
New meta-scientific review questions the evidence for the gold standard psychotherapies and empirically supported treatments.
Pervasive Industry Influence in Healthcare Sector Harms Patients
Experts across the globe point to the harms of drug companies’ influence on research, practice, and education in healthcare noting that it compromises patient care.
Statisticians: Current Policies Approve Ineffective Treatments
Current standards for clinical trials rely on statistical methods that allow for ineffective treatments to gain approval.
Researchers Address Dangers of Polypharmacy and Inappropriate Medication Use
A new special issue brings together articles exploring the harmful effects of simultaneous multiple medication use.
D-Cycloserine Supplement Does Not Add Much to Exposure Therapy
A closer look at a new study reporting that the supplement D-cycloserine improved anxiety when used with exposure therapy.
Despite Claims, EPA Supplement Does Not Improve ADHD Symptoms in Youth
A new study reports that the supplement EPA improved ADHD symptoms but a closer look calls these results into question.
Textbooks Provide Misleading Information on the Neurobiology of ADHD
When it comes to ADHD, some researchers suggest that medical textbooks provide inaccurate and misleading information.
NICE Guideline Update Acknowledges Severe Antidepressant Withdrawal
A new update to the NICE guideline for depression suggests providers discuss long-term, severe antidepressant withdrawal symptoms.
“Flimsy Evidence” for Esketamine as Depression Treatment
A new article documents the “flimsy evidence” behind the recent FDA approval of the party drug esketamine for the treatment of depression.
Researchers: “Antidepressants Should Not be Used for Adults with Major Depressive Disorder”
A new review, published in BMJ Evidence-Based Medicine, concludes that antidepressants should not be used as the risks outweigh evidence for benefits.
Zoloft Does Not Improve Depression, Even in Severe Cases, Study Finds
Despite their finding, the researchers suggest that SSRIs be given to people who do not meet criteria for depression or anxiety.
Guidelines Recommending Antidepressants “in Contradiction with the Current Evidence”
Researchers critique the German S3 guidelines for depression promoting antidepressants.
Substantial Spin Found in Leading Psychology and Psychiatry Journals
: A new review finds evidence of spin and the misrepresentation of clinical trials with non-significant results.
Review Finds FDA Approval of Digital Antipsychotic Misguided
The approval of the digital antipsychotic may open the door for more pharmaceutical company profits without evidence of benefits to patients.
Positive Antidepressant Study “Misleading” and “Erroneous”
An analysis of last year’s positive finding in The Lancet about antidepressant efficacy shows errors, obfuscations, and misrepresentations.