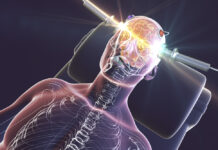
In December, the FDA proposed down-classifying the device used in electroshock treatment (ECT), which would loosen regulations and potentially make ECT more widely available for use. In response, five individuals who were previously harmed by electroshock have filed a Citizens Petition with the FDA.
“I hope this is the beginning of the path to do away with shock [ECT]. Hopefully, it’ll go the way of lobotomies, another ‘miracle treatment’,” one ECT survivor, shocked as a teenager, said in a release.
More:
Full press release: http://www.prnewswire.com/news-releases/attorney-jonathan-emord-takes-on-the-fda-over-electroshock-ect-device-300318482.html
A copy of the Petition is available here, along with supporting Exhibits (Part 1, Part 2, and Part 3).
Note: This initiative may be associated with CCHR