Tag: RCT
A New Paradigm for Testing Psychiatric Drugs Is Needed
This paper reviews the problems with the usual double-blind, placebo-controlled trials on which drug approvals are based, and advocates for a stricter form of testing psychiatric drugs with patient-relevant outcomes, real comparators, long-term outcomes, and assessment of harms.
Research Progresses on Mindfulness Based Interventions for Adolescents
A new meta-analysis analyzes randomized control trials of Mindfulness-Based Interventions for adolescents.
Peer Support Reduces Chances of Psychiatric Readmission
A randomized control trial finds that receiving peer support from individuals with similar lived experiences reduces one’s risk of readmission to an acute crisis unit.
Prominent Researcher and Psychotherapist Questions “Evidence-Based Therapy”
Dr. Johnathan Shedler recently published a paper critiquing how the term “evidence-based” is being used in the field of psychotherapy.
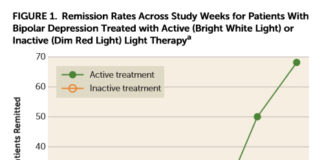
Bright Light Therapy More Effective Than Medication Alone for Bipolar Depression
A new randomized, double-blind, placebo-controlled trial has found bright light therapy to be a powerful intervention that could provide an alternative to medication for people with “bipolar depression.”
Vitamin B6 Effective in Reducing Antipsychotic Induced Akathisia
A recent RCT showed that vitamin B6 is as effective as propranolol for the treatment of akathisia.
Smartphone Based Interventions for Depressive Symptoms
New meta-analysis of smartphone based interventions demonstrates small-to-moderate effect.
Nominee to Lead FDA Removed Name From Recent Publications
Sheila Kaplan for the Boston Globe reports that Dr. Robert Califf, the Obama administration's nominee to lead the Food and Drug Administration (FDA), has removed his name from a series of scientific papers that he recently coauthored. The decision to remove his name, against publication ethics standards, has brought Califf under renewed criticism.