Over the past twenty years, the number of prescriptions for atypical antipsychotics written to children and young adults between four and eighteen has increased precipitously, according to the results of a recent study published in the journal Medicine. Two-thirds of these antipsychotics were prescribed “off-label” for unapproved uses, many for children diagnosed with ‘ADHD.’
“It’s concerning that such a high proportion of pediatric patients were taking [atypical antipsychotics] AAPs off-label and that a number of children are exposed to AAP side effects such as metabolic syndrome and diabetes, while the drugs’ effectiveness on their psychiatric condition may not have been well-established,” Minji Sohn, a co-author of the study, told Contemporary Pediatrics in an interview.
Antipsychotic drugs have received FDA approval for only schizophrenia, bipolar mania, irritability associated with autism, and Tourette syndrome in children. When atypical antipsychotics were introduced during the 1990s, however, they were marketed as having fewer side-effects than the first-generation antipsychotics that preceded them. As a result, it became increasingly common to prescribe these drugs for conditions other than those for which the FDA had approved.
The practice of prescribing antipsychotics for unapproved conditions, also known as “off-label” prescribing became controversial, however, as post-marketing research found the potential for serious adverse effects, including metabolic syndrome, cardiovascular complications, and even death. “Nevertheless,” the researchers point out, “AAPs are one of the top-selling classes of pharmaceuticals in the United States.”
“In fact, antipsychotic medications generated about $18.2 billion total revenue in 2011, with 3 individual AAP agents accounting for 65% of the total revenue. In children and adolescents in the United States, AAPs are probably among the most increasingly used classes of prescription drugs.”
The present study attempts to examine the historical trend on antipsychotic prescribing to youth in the US, specifically to those diagnosed with ‘ADHD’ who are being prescribed these drugs off-label. Using data from the National Ambulatory Medical Care Survey (NAMCS) and the National Hospital Ambulatory Medical Care Survey (NHAMCS), the researchers looked at national prescribing trends between 1993 and 2010.
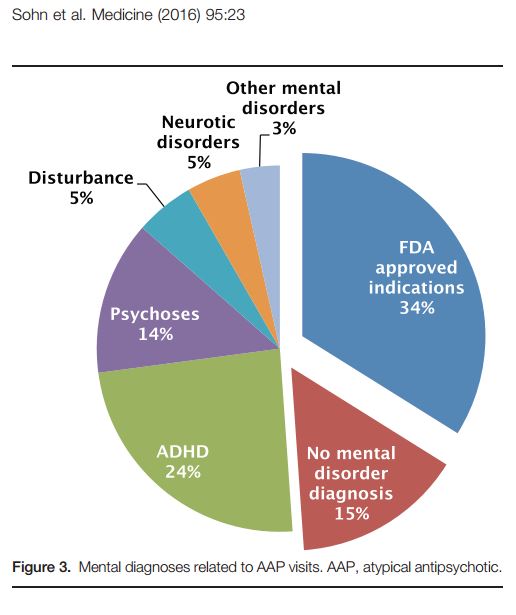
They found that throughout the study period the majority of children and youth receiving antipsychotic drugs did not have a one of the FDA approved conditions. Only 34% of those receiving prescriptions were diagnosed with one of the conditions for which the FDA had approved the drugs use, with the majority of patients having only a diagnosis for ‘ADHD’ (24%).
Of all of the children going in for a pediatric appointment concerning an ‘ADHD’ diagnosis, 12% received at least one prescription for an atypical antipsychotic. Patients with Medicaid as a primary source of payment were significantly more likely to receive an antipsychotic prescription for ‘ADHD.’
“Our analysis for identifying mental diagnoses that are related to AAP prescriptions revealed that approximately 66% of total pediatric AAP visits did not include a diagnosis for US FFDA-approved indications,” they conclude. “Of those, ADHD was the most common primary mental diagnosis.”
Overall, the use of atypical antipsychotics increased steadily with large increases between 1999 and 2000, when the rate doubled, and between 2002 and 2003, when the rate doubled again (from 0.8 per 100 visits to 1.6 per 100 visits).
****
Sohn, M., Moga, D.C., Blumenschein, K. and Talbert, J., 2016. National trends in off-label use of atypical antipsychotics in children and adolescents in the United States. Medicine, 95(23), p.e3784. (Abstract)















Why are things like this no longer surprising? And in fact we have come to expect them. What are we going to do about it?
Report comment
So when are people going to superglue themselves to pharmacy counters?
Lets remember when children show signs of distress there are reasons.
Lets remember when people of all ages are drugged for showing signs of distress we let the abusers off the hook.
Report comment
Well, they won’t be hyperactive when they’re 50+ pounds overweight, particularly when they’re only in elementary school. Who says psychiatrists don’t have brilliant minds?
Report comment
The entire trend is ridiculous and very concerning. Antipsychotics deplete critical nutrients too: http://www.optimallivingdynamics.com/blog/7-important-nutrients-depleted-by-psychiatric-drugs-antidepressants-antipsychotics-stimulants-benzodiazepines-induced-guide-vitamins-medications
Report comment
More kids getting anti psychotics cause they are giving them that “long acting” Vyvance and are bugging out and can’t sleep.
That Vyvanse pro drug delivery system is flawed, that amphetamine feeling peaks real high then the only “long acting” part of the deal is the anxiety ridden can’t eat feel bugged out crash and can’t fall asleep. That crash part basically is “psychosis”, you are just not right.
http://www.google.com/search?q=vyvanse+crash < Like a thousand stories describing it.
Old school Adderal IR would get you in the 'get er done' zone for a couple of hours then a moderate crash with that Vyvanse you get the same few hours but that crappy jittery anxiety cant eat its still in me but not working part just goes on and on and on crashing bugged out for hours.
Shire doesn't give a crap their new product is horrible , new patent = billions in revenue .
So anyway of course as more kids get the new make you bugged out all day and into the night Vyvance more kids are getting anti psychotics to counter act it.
Report comment
There clearly needs to be a major campaign to educate the general public about the fact that besides that psychiatric drugs are approved for “conditions” on the basis of poorly conducted and often dishonest research that people, including children, are being massively drugged for “conditions” for which these drugs were never approved. The question is: Where does one get money to fund such a campaign?
Report comment
Don’t forget the brilliant move of introducing addictive antipsychotics for general use, like we have now.
Report comment
A good point. We could actually get a lot of mileage out of simply pointing out that they don’t even follow their own research based on their own idiotic diagnostic protocols and categories. Within their own paradigm, giving antipsychotics for ADHD would be like giving blood pressure medication for someone with back spasms. I have no idea how they get away with it, really. Mass hypnosis, I guess.
As for funding, that’s the rub, isn’t it?
—- Steve
Report comment
Many, many of the cases where “antipsychotics” are prescribed for “ADHD” are a result of kids becoming aggressive as an adverse effect of the stimulants they’ve been prescribed. I see this regularly in my work advocating for foster youth.
The truly bizarre nature of this approach is only apparent if you know a little of the biochemistry involved. Stimulants increase levels of dopamine available in the brain, which can cause aggression, especially in those who have a trauma history. Antipsychotics DECREASE the same chemical (dopamine) that the doctor has been INCREASING with the stimulants!!! So they raise dopamine with one drug while suppressing it with another. And our tax dollars pay for this stupidity????
I don’t know why people are so willing to allow this chicanery to pass unnoticed!
—- Steve
Report comment
It breaks my heart doctors are forcing these toxic neuroleptics onto children, since torturing children is not actually appropriate medical care.
http://www.narpa.org/UN-forced%20treatment%20is%20torture.htm
Although, now that all the DSM disorders are known to be scientifically invalid and unreliable:
http://www.nimh.nih.gov/about/director/2013/transforming-diagnosis.shtml
Does it really matter what diagnosis the children are given? The reality is no child should be given antipsychotics. Period. Prescribing antipsychotics to children is child abuse.
Report comment