Andrew Yount of Tennessee was a four year old with behavior problems and an ADHD diagnosis in the summer of 2003 when his doctor prescribed Risperdal.
The drug was a powerful adult antipsychotic marketed aggressively for children by Johnson & Johnson to meet the pharmaceutical company’s billion-dollar-a-year sales goal.
By Christmas, after Andrew had taken Risperdal for four months, holiday photos show that this preschool boy had developed breasts like those of a young girl in the beginning stages of puberty.
The doctor who prescribed Risperdal to Andrew did not know what J&J studies showed: that the drug raised levels of the hormone prolactin which triggered the growth of female breasts, known as gynecomastia, in a significant percentage of boys who took the medicine.
And once the growth began, there was no going back.
“The match has been lit to light the fire, and the cells have started doing what they’re going to do. They are going to continue to grow disproportionately to the rest of him,” Mark Solomon, a medical expert on Risperdal’s gynecomastia side effect, testified about Andrew in a Philadelphia courtroom in June 2016.
Today, at 18, Andrew is a fully grown man with pendulous, 42D-size breasts.
And he’s not the only one. Thousands of boys and young men are lined up in courthouses around the country to sue J&J for gynecomastia caused by taking Risperdal as young children. The condition is irreversible except by surgical removal.
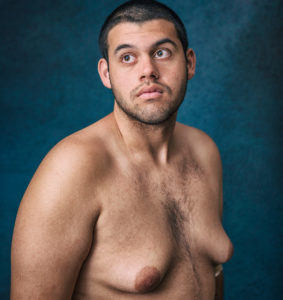
Collectively, they have become known as the Risperdal Boys after a handful of the now-grown men agreed to participate in a photo shoot and interviews to help get their story out. The result is posted at risperdalboys.com.
Some of the Risperdal Boys have won settlements and verdicts from J&J, including a $76 million judgment in the Yount case in 2016. His is the biggest payout to date. The verdicts are based on the question of whether the medical community was adequately warned about the gynecomastia side effects of Risperdal.
J&J strenuously denies liability and insists consumers were adequately warned about side effects.
The Risperdal Boys are victims of an enormously profitable strategy used by J&J and other drug companies to increase revenue by getting doctors to prescribe their drugs to patients for whom the drug was never intended. It’s called prescribing drugs “off-label,” and it means that patients are turned, in essence, into guinea pigs by taking a drug which was never proven to be safe or effective for their medical condition.
That the pharmaceutical companies are driven more by profit than compassion is evidenced by the lack of follow-up by the companies to determine whether these off-label patients benefited or were harmed by the drug.
In the case of Risperdal, FDA approved the drug in 1993 to treat adult schizophrenics. But the percentage of schizophrenics in the U.S. population is too small for the drug to make a lot of money by Johnson & Johnson’s standards. So in the late 1990s and early 2000s, the J&J marketing team focused on convincing doctors to prescribe Risperdal to tens of thousands of young children with a variety of behavior issues, according to testimony.
While it’s illegal for drug companies to market a drug for a group not approved by the FDA, doctors are free to prescribe drugs as they see fit.
The FDA had explicitly rejected several requests by J&J to approve the drug for children because the company didn’t have sufficient data to show it would be safe, according to testimony by former FDA commissioner David Kessler.
According to the online legal news publication Law 360, Kessler also testified in court that “about 20 percent of the Risperdal prescribed before the early 2000s was used in children and adolescents.”
In addition to children, J&J encouraged doctors to prescribe Risperdal off-label for elderly people with dementia, according to the U.S. Justice Department. Risperdal did not have FDA approval for use in the elderly, and these people were later found to have suffered increased risk of strokes after taking the drug.
J&J paid $2.2 billion in 2013 to settle criminal and civil claims by the U.S. Department of Justice that it paid kickbacks to doctors to prescribe Risperdal and two other drugs off label and to get those doctors to persuade their medical colleagues to do the same. In the settlement, J&J paid criminal fines and forfeiture for violations of the law and civil settlements based on the False Claims Act, according to the DOJ. The case was one of the largest health care fraud settlements in U.S. history. Whistleblowers from within the company had tipped off the feds.
“J&J and Janssen knew that Risperdal posed certain health risks to children, including the risk of elevated levels of prolactin, a hormone that can stimulate breast development and milk production. Nonetheless, one of Janssen’s Key Base Business Goals was to grow and protect the drug’s market share with child/adolescent patients. Janssen instructed its sales representatives to call on child psychiatrists, as well as mental health facilities that primarily treated children, and to market Risperdal as safe and effective for symptoms of various childhood disorders, such as attention deficit hyperactivity disorder, oppositional defiant disorder, obsessive-compulsive disorder and autism,” the DOJ stated in a press release.
That same year, the company reported total worldwide sales of $71.3 billion, according to a company press release.
If billion-dollar penalties are being factored in by the drug industry as somehow an acceptable cost of doing business, the calculation fails to take into account the cost of irreparably damaged lives.
The damage to the Risperdal Boys is not only physical but emotional and psychological.
As early as age six, Andrew was refusing to take off his shirt in front of his own doctor.
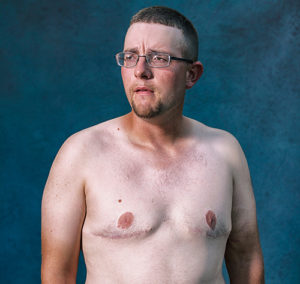
Eddie from Oklahoma, who participated in the photo documentary, described how the bullying led him to withdraw into a lonely world playing alone at home in front of a video screen.
“I had bigger boobs than most of the girls in high school,” Eddie said on the Risperdal Boys documentary.
He was fortunate that Oklahoma’s SoonerCare paid for his mastectomy.
Some of the Risperdal Boys describe trying to bind their breasts to make them less noticeable, and not finding anyone to date. After being groped first by a girl and then a boy in class, Arturo from Pontiac said he left school and never went back.
Because J&J had downplayed the connection between Risperdal and gynecomastia, Isaiah from Flint went through tests for cancer before his doctors, trying to find an answer for his unusual breast development, discovered his elevated levels of prolactin.
Perhaps one of the best known of the Risperdal Boys is Austin Pledger, a young Alabama man in his early 20s. His mother Benita spoke for him in court during his trial against J&J. Austin’s family won the first jury verdict against J&J — a jury in Philadelphia awarded Austin $2.5 million.
Austin’s mother Benita recalls filling his first prescription for Risperdal in 2002, when he was eight years old, as treatment for irritability associated with autism. The doctor who prescribed Risperdal to Austin had received the full Risperdal marketing treatment from the local sales representative for J&J’s subsidiary Janssen. The sales rep visited the doctor at least 20 times, twice as often as Austin saw the doctor, according to testimony.
Austin stayed on Risperdal for five years. By the time he was an adolescent, Austin had full female breasts.
At trial Benita described how Austin would put a towel over his breasts when he got out of the shower so he wouldn’t have to see them in the mirror, and how he intentionally banged his breasts on a table.
J&J acknowledged in its 2016 annual report that the company is facing 18,500 personal injury lawsuits worldwide relating to Risperdal. Many of the boys need help to pay for costly plastic surgery to undo the damage from the drug.

Of all the Risperdal claims filed against J&J, at least three are known to have been settled by the company on the eve of trial.
Since Yount’s case, judges in Philadelphia’s mass tort court have stopped four cases from reaching a jury on technical grounds. All four decisions are on appeal. J&J is also appealing verdicts against the company.
There will be more Risperdal cases in the coming months.
The new cases will bring the kind of publicity that J&J doesn’t need, coming off recent weeks of troubling headlines. Bloomberg reported J&J lost six of the seven largest product liability verdicts in 2016. Besides the $76 million Yount judgment, juries gave two hip implant patients a total of $1.5 billion, and three women alleging the company’s talc caused ovarian cancer got a total of almost $200 million. Some of the verdicts were reduced by judges, and some are expected to be appealed. The company acknowledges more than 100,000 product liability lawsuits pending, including cases involving allegations that its blood thinner Xarelto causes uncontrolled bleeding and its pelvic mesh deteriorates and causes internal damage. Bloomberg reported that 17 product liability cases against the company involving death and injury were expected to go to trial in 2017.















It’s been proven Risperdal causes brain shrinkage, too. Where are the lawyers in regards to the fact that Rispedal causes brain damage? Not to mention it makes people “psychotic,” via anticholinergic toxidrome poisoning, while it’s falsely advertised as an “antipsychotic.”
https://en.wikipedia.org/wiki/Toxidrome
I hope J&J loses it’s shirt due to these Risperdal lawsuits, just like the Risperdal boys.
Report comment
Until CEO’s and upper managerial staff of drug companies start going to prison for this kind of crime against humanity nothing is going to change. The drug companies make so many billions in profit from their off label promotions of their poisons that monetary awards mandated by the courts will remain only the cost of doing business. They build money into their budgets for taking care of the fines imposed on them by the courts. Most of this would stop if people started going to prison.
Drug companies do not care about how their products harm the lives of countless individuals.
Report comment
Stephen,
I think the drug companies are in a sense more powerful than the regulators or the government. I think if any politician steps out of line and becomes critical – they can be ‘got’.
Report comment
As long as they depend on drug companies for their campaign funds, you’re right. That’s why campaign finance reform, in the form of banning ALL corporate contributions to candidates, is the issue of our age.
Report comment
What Mr. McCrea said, but also “got” in another way. A drug company’s sales staff is charged with forming relationships with doctors. A plausible conjecture: A few well-funded drinking sessions with legislators’ doctors in Washington, DC will build enough misplaced trust that a doctor of weak character violates HIPAA to get a few laughs or feel like a bigshot. A doctor waking up the morning after a night of debauchery with fun-loving sociopaths from Johnson & Johnson might not remember disclosing medical information about high-profile patients, but the J&J representatives remember every word.
Report comment
Thank you for this report. The business practices of Big Pharma are more than familiar to me, but I hadn’t been following this particular story closely.
Report comment
A truly fine drug. Abram Hoffer, who invariably tried to get his patients off psych drugs, also found it to be addictive, as well.
Report comment
Somehow this topic (risperdal-induced male breast growth) is new to me and I’d thought I’d heard it all. The images are shocking. What’s even more shocking is that the effects of the drugs are called ‘side’ effects. Can we just call this one of the actual effects of intentional poisoning once and for all? If J&J knew this was an effect, why do doctors and parents need to be warned about the risk? Why hasn’t the drug simply been withdrawn and acknowledged as a dangerous substance with no value except to those whose only goal is a zombified “compliant” child? When are the doctors and researchers going to acknowledge that in many cases, changing the parents behaviors would have more desirable long term effects than pathologizing the child’s reaction to trauma and dysfunction?
Report comment
Yes, and i have proof that this was known, reported and refused to be addressed. After 3 years of begging and crying, I was taken to a Endricrinologist who said they should have stopped the drugs and that if I saw him sooner he would have prescribed Synthroid, to try to stop it.
I remember the other prisoners taking polaroids of me in the shower and showing everyone, including all the girls, who would grab my “tits” and compare me to them and then go sleep with the other male residents in the same room where I first showed the Psychiatrist my “tits” and begged for help. Also the same room where I was forced to have my first sexual experience. I was always reminded of my inferiority and uselessness.
This same room was the Psychiatrists office.
All for the sake of covering up abuse that they documented was occuring and that I was being victimized. Money talks!!!
Report comment
The current medical director of Riverside in Massachusetts fully knew that the pills he gave me made me grow breasts. No one cared about the damage this did to me. My first sexual experience was forced on me in his office . My rectum was penetrated 7 times because of this doctor. All of this before the age of 16. This is despite my medical records from this time period stating that I was not “mentally ill ” and this doctor himself openly stated and had documented, that in fact, he knew that I was abused my family and psychiatry was their tools of abuse. I have a written document in form a transcript written by clinicians of a phone call in which my parents called screaming and threatening action against the doctor if he did not change my medical records from stating that my parents were “mentally ill “and that I was ” healthy” to “tell ****** that he is mentally ill and that ******* are healthy “. I lost 30 Years of my life because of a documented lie and I am still devastated by it. Thankfully I had plastic surgery but i never recovered. F*** Mad in America for repeatedly denying my voice form being heard. Whoever is running Mad in America has not responded to several attempts over the years to allow someone in its own backyard a chance to be heard. This website allows numerous survivors and advocates their voice, yet, somehow despite having hundreds of documents with all the names of “providers”, all Massachusetts based and many still working today, with a few of them working alongside Psychiatric Survivors . Why does Mad in America refuse to allow some people a voice? I read articles often on this website often, written by the haves, while have-nots in your own backyard, aren’t granted a single response in 5 years of requesting a chance to post a blog . Mad in America – stop silencing people!
Report comment
Documentation proves that upon being threatened, the medical records were changed
Report comment
Forced Gynecomastia stole my adolescence from me. Forced rectal penetration all by women. The humiliation and the torment was endless. I have never been ok since. I don’t eat regularly and sometimes have to do things that most reading this would want to end their life if they had no other options to get basic needs met.
As I have been told countless times; it’s my fault.
Report comment
There’s nothing as devastating as a family in league against one child, except a family clever enough to weaponize its malevolence with doctors and drugs. Hardships endured with the support of a loving family pale in comparison. And, of course, there’s no one more vulnerable or less trusted than someone who has been diagnosed with a mental illness and made to take drugs. The drugs validate the diagnosis, which after all is just words, by introducing physical objects that can be seen and touched. They also cause mental states that match the signs of various psychiatric diagnoses and harm physical health in ways that can be attributed to the child/victim’s supposed “mental illness.” Obesity, anorexia, tics, lethargy, insomnia, somnolence, mania, depression, and hallucinations, all caused by drugs and all presumed to be caused by “emotional problems.” Between the meaning of drugs and their disabling effects, they’re self-refilling prophecies.
Your upbringing was a long-term crime that went undetected because the bad guys had the field of medicine as their cover, but your ability to write about it casts you in more roles than mere victim. You can be a detective, a prosecutor, a judge, and a journalist. Those are powerful positions from which you can correct the lies and reveal the villains. If you can’t find an outlet for your history here or on another site, you can publish it yourself on Medium.com and promote it on Twitter and Facebook. You can self-publish a book that would be printed on demand or distributed in digital form, with revenue sharing but no up-front costs, on Amazon.
Too many people are completely ignorant of the grievous harm visited upon innocent individuals like you in the name of “mental health care,” so every effort to inform is worth making. More power to you.
Report comment
Yeah, most guys don’t want to grow breasts, and I’m thankful for this lawsuit to stick it to the drug company, but where is the lawsuit for metabolic syndrome (diabetes and cardiovascular disease caused by all neuroleptic drugs)? Growing breasts would be embarrassing, but metabolic syndrome is quite deadly. And yes, these people need jail time, not just fines.
Report comment
But…
But nothing.
Embarrassing? That is how you think it feels. It is similar to having a crumb on your cheek and no one telling you. Very embarrassing.
You think this lawsuit sticks it to the drug company? They don’t care about me or you. That is how much money they have.
Report comment
I have been on valproate for long and have gained more than 15 kg,developed alopecia,diabetes,hypertension.Then they substituted it with carbamazepine and immediately my hormone level dropped and I have to take testosterone every month and I also got a divorce immediately after that.
Do you think I can take valproate and CBZ to the court?Valproate and CBZ interaction is mentioned in the literature.
Report comment
Good that the J and J continues to be sued for the devastation caused, though the real issue has not been addressed and that is justice. As others have said here on the thread, the behavior won’t change until the individual executives face prosecution. Over at Eli Lilly, Mitch Daniels, in charge of the roll-out of Zyprexa as head of North American operations, went on to become Governor of Indiana and is now President of Purdue University. And, because these executives make group decisions (in Lilly’s case, to ignore, deny, etc., the fact that Zyprexa causes diabetes, hyperglycemia, and death – even as they were told so by a panel of expert endocrinologists) they are able to sleep at night with a clear conscience. At least that’s how I imagine it.
Report comment
This is a much bigger scandal than this journalist describes, and I have two concerns about this article, although I am of course glad that yet another article about the Risperdal scandal has been written. One concern is why the “marketing group” to which the writer refers is not identified, why this writer — like every other major media person who has written about the scandal — fails to mention the readily available information about the three psychiatrists who were paid vast sums of money to create a false “Practice Guideline” for using Risperdal that laid the very foundation for all that followed AND who also were paid vast sums after that to produce what their leader, Allen Frances of DSM-IV fame (infamy) called their “marketing plan.” Why do the major media people protect these people over all these years, when they readily name the names of others who were involved. The full story is told in this video https://www.youtube.com/watch?v=tgilBaRbulc and in these articles, one of which is in MadinAmerica: http://www.oa.uottawa.ca/journals/aporia/articles/2015_01/commentary.pdf and https://www.madinamerica.com/2015/10/diagnosisgate-a-major-media-blackout-mystery/
My other concern is about how the writer of this article, like investigative Stephen Brill in his 15-part series (!) about the Risperdal scandal, focus so intensively on gynecomastia. Certainly the photos that accompany the current article are intended to be shocking, but I would not have expected MadInAmerica to decide to include any such photos, never mind several! I do feel badly for the young males who were already suffering in some way — or causing trouble for others — because why else would they have come to the notice of whoever put them on Rispderal? And on top of whatever else was going on, to be adolescent boys or men and develop breasts must have been terrible. But it is terrible because of the combination of misogyny — “Eww, who wants to have a body part that is associated with women?!!” — and the rigidity, manifested in so many ways, that people have about sex and gender. As some of the other people who commented on this article have wisely observed, where is the alarm about the other Risperdal-caused harm, including shrinkage of the brain, diabetes, hyperglycemia, and even death? Why is this entire article and a 15-part series focused on gynecomastia?
Report comment