Can a Conceptual Competence Curriculum Bring Humility to Psychiatry?
Training for conceptual competence in psychiatry provides a new way forward to address theoretical and philosophical issues in mental health research and practice.
How Dissenting Voices are Silenced in Medicine
Researcher criticizes the many ways opposing viewpoints and dissenting voices are squashed in the field of medicine.
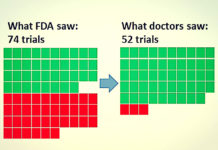
Where Are the Results of These Five Clinical Trials of Antidepressant Drugs?
The results of five large-scale clinical trials of antidepressants have never been made accessible to the public, a data set compiled by an international team of researchers shows. Their discovery highlights the incompleteness of available data on the safety and efficacy of antidepressant drugs.
Researchers: Antidepressant Withdrawal, Not “Discontinuation Syndrome”
Researchers suggest that the pharmaceutical industry had a vested interest in using the term “discontinuation” in order to hide the severity of physical dependence and withdrawal reactions many people experience from antidepressants.
Amsterdam Files New Study 352 Whistleblower Complaint
Jay Amsterdam, who first blew the whistle on corrupt research practices in a study conducted by GlaxoSmithKline (GSK) eight years ago, has now submitted...
Biogen Pushes FDA to Approve Failed Alzheimer’s Drug
A new analysis, published in Lancet Neurology, demonstrates how Biogen is spinning results from two failed trials for a new Alzheimer's drug.
Researchers Set the Record Straight on Controversial Zoloft Study
An issue of Lancet Psychiatry is devoted to clarifying the lack of efficacy for Zoloft (sertraline).
Pervasive Industry Influence in Healthcare Sector Harms Patients
Experts across the globe point to the harms of drug companies’ influence on research, practice, and education in healthcare noting that it compromises patient care.
Researchers Critique WHO Mental Health Technology
Researchers critically examine the underlying assumptions and implications of a new WHO mental health technology designed to streamline psychiatric assessment internationally.
Statisticians: Current Policies Approve Ineffective Treatments
Current standards for clinical trials rely on statistical methods that allow for ineffective treatments to gain approval.
Researchers Address Dangers of Polypharmacy and Inappropriate Medication Use
A new special issue brings together articles exploring the harmful effects of simultaneous multiple medication use.
D-Cycloserine Supplement Does Not Add Much to Exposure Therapy
A closer look at a new study reporting that the supplement D-cycloserine improved anxiety when used with exposure therapy.
Textbooks Provide Misleading Information on the Neurobiology of ADHD
When it comes to ADHD, some researchers suggest that medical textbooks provide inaccurate and misleading information.
“Flimsy Evidence” for Esketamine as Depression Treatment
A new article documents the “flimsy evidence” behind the recent FDA approval of the party drug esketamine for the treatment of depression.
Researchers: “Antidepressants Should Not be Used for Adults with Major Depressive Disorder”
A new review, published in BMJ Evidence-Based Medicine, concludes that antidepressants should not be used as the risks outweigh evidence for benefits.
Zoloft Does Not Improve Depression, Even in Severe Cases, Study Finds
Despite their finding, the researchers suggest that SSRIs be given to people who do not meet criteria for depression or anxiety.
Substantial Spin Found in Leading Psychology and Psychiatry Journals
: A new review finds evidence of spin and the misrepresentation of clinical trials with non-significant results.
Review Finds FDA Approval of Digital Antipsychotic Misguided
The approval of the digital antipsychotic may open the door for more pharmaceutical company profits without evidence of benefits to patients.
Getting Pharma Out of Medical Education: An Interview with Dr. Adriane Fugh-Berman
MIA's Gavin Crowell-Williamson interviews PharmedOut founder Dr. Adriane Fugh-Berman about Big Pharma's influence on medical education.
Psychotropic Medications Serve as Powerful Tools for U.S. Military, Imperialism
Ethnographic research sheds light on extensive psychopharmaceutical use by soldiers in post 9/11 U.S. wars.
Mobile Apps for Mental Health Lack Transparency in Data Sharing
Research illustrates privacy concerns with how mental health applications collect and share users’ data.
Fighting for the Meaning of Madness: An Interview with Dr. John Read
Akansha Vaswani interviews Dr. John Read about the influences on his work and his research on madness, psychosis, and the mental health industry.
Researchers Find Bias in Industry-Funded Continuing Medical Education
Industry-funded continuing medical education (CME) influences physicians to prescribe more opioids, focus less on the consequences.
Researchers Fail to Replicate Evidence for “Biotypes” of Depression
A new study casts doubt on whether such biotypes for depression exist.
Antidepressant Use Linked to Longer, More Frequent Psychiatric Rehospitalization
New study finds that antidepressants may negatively impact recovery after psychiatric hospitalization.