I’d like to comment further on the issue of Deep Brain Stimulation (DBS), partly to clarify a couple of issues I should have addressed at more length in my last post, and partly to draw attention to a deeply alarming example of experimentation on patients that surfaced in the Washington Post this week.
The recent report on DBS published in the American Journal of Psychiatry this month was an open, unblinded study undertaken in the aftermath of a catastrophically bad clinical trial that was blinded. Not only did that trial (and another parallel trial sponsored by a competing medical device manufacturer) fail to show any efficacy in treatment-resistant depression, it exposed the participants to a range of extremely serious, even life-threatening complications. Undaunted, Helen Mayberg and her colleagues running her failed trial left the devices in place in the patients who could tolerate them, and continued to stimulate their brains.
Because of the massive conflicts of interest that plague academic medicine, some years back medical journals began to require that authors of papers declare financial ties that might call into question the independence of the authors’ assessment of the trials they were reporting on. These new rules have proved to be little more than a fig leaf, masking the ways in which sponsorship by interested parties leads to “findings” that are heavily biased.1 When comparisons are made between studies of drugs sponsored by Big Pharma, and trials sponsored by federal dollars, those with drug company sponsorship routinely report far more favorable results than studies where no commercial interests are at stake.2 The situation is even worse when it comes to medical devices. A study appearing last year in JAMA Surgery, “Association of Compensation from the Surgical and Medical Device Industry to Physicians and Self-Declared Conflict of Interest,” examined whether the 100 physicians receiving the highest compensation from 10 large surgical and medical device manufacturers actually declared their conflicts of interest. The results were deeply disturbing, though not surprising: Conflicts of interest were declared in only 84 of 225 relevant publications, or 37.3%. In other words, in nearly two-thirds of cases, these physicians lied, and of course they did so with impunity. So it is that “evidence-based medicine” becomes “evidence-biased medicine.”3
But serious problems exist even when possible conflicts of interest are “declared.” The relevant statements at the end of academic papers provide minimal detail, thus disguising the true extent of the conflicts that exist. If we know only that X received some grant support from company Y, we are left in the dark about the extent of this support. In the case of the recent American Journal of Psychiatry paper, not only were the authors indebted financially to the medical device manufacturer seeking to expand the market for its wares, but at least one of the authors, Helen Mayberg, owned patents covering the devices in question. Were results to be seen as favorable, the potential financial and career rewards for her (and for the others participating in the study) were extensive and readily overlooked by those reading her study, given the imprimatur of one of psychiatry’s two leading professional journals.
It should come as no surprise that, in the face of clear negative findings from two controlled trials, these authors were nonetheless tempted to continue to make a case for their experimental treatment, despite unimpeachable evidence of the harm suffered by many of their experimental subjects (as I summarized in my previous blog piece). That they should have been allowed to do so is a scandal. That an accompanying editorial in the American Journal of Psychiatry should laud them as examples of “rigorous clinical research efforts” and “outstanding examples of the efforts and dedication of researchers committed to helping clinicians successfully treat patients whose symptoms are not responsive to treatment” leaves one close to despair. The cherry-picked data in the paper by Mayberg et al. should give any moderately critical reader pause, and the fact that positive outcomes only appear when biased researchers are allowed to self-assess the results of their interventions is telling. And that is to leave aside the often extremely serious sequelae associated with this form of psychosurgery.
When I published a study of an earlier example of the desperate remedies to which mental patients have often been subjected (removal of teeth, tonsils, and internal organs as part of an attempt to extirpate the focal sepsis that was allegedly poisoning mental patients’ brains, killing hundreds and maiming thousands, an outcome covered up at the highest levels of the profession), I was often asked if something similar could happen today. My response, which present events vindicate, was that though history rarely repeats itself exactly as before, similar horror stories could not be ruled out. Mental patients remain extremely vulnerable. The etiological understanding of mental disorders remains strikingly primitive, and such treatments as we have provide at best a measure of symptomatic relief to a subset of these patients, often at a very heavy price in terms of side effects. Ambitious clinicians let loose in such a situation can easily be carried away by their enthusiasms, and the restraints on experimentation, while stronger than they once were, remain inadequate.
One further remark before I pass on to this week’s news: when lobotomy was introduced in the mid-1930s, it was popularized by a duo consisting of a neurologist (Walter Freeman) and a neurosurgeon (James Watts). The vogue for DBS is to some degree the result of the self-promoting activities of another duo, also a neurologist and a neurosurgeon. Both groups, of course, needed the active collaborations of some psychiatrists to advance their work, but the impetus came from outside psychiatry itself. And the work of the lobotomists and of the latest advocates of a deeply invasive neurosurgery was in both instances greeted by uncritical articles by science journalists who should have known better. I am constantly appalled by articles even in serious newspapers that breathlessly tout the latest medical “breakthrough” — based on an anecdotal study or two that proves precisely nothing.
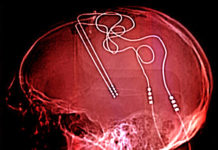
Which brings me to this week’s example of the genre, which appeared in the pages of the November 6th issue of the Washington Post. A fascinating and immensely promising new treatment was being advanced as a breakthrough in the treatment of the scourge of opioid addiction. And what was this miraculous new intervention? Implanting electrodes deep in the brain, and using a battery implanted elsewhere in the body to zap the addict and keep him from relapsing.
The reporter regaled us with the story of a heroic neurosurgeon who implanted this device after seven hours of neurosurgery. It was, we were solemnly assured, akin to the medical miracle that is a heart pacemaker. This one was a brain pacemaker, rejiggering the functions of the brain. Think about that for a second — a heart pacemaker has a relatively simple, if vital, task to perform: causing the muscular pump that is one’s heart to maintain a steady beat in cases where it is prone to beat irregularly or too slowly. The human brain is not a pump.It is a fantastically complicated organ composed of about 200 billion nerves cells, interconnected in hundreds of trillions of ways. Despite all the progress of neuroscience over the past half-century, our understanding of how it is put together, and how all those myriad connections function, remains strikingly primitive. The idea of a “brain pacemaker” is so ludicrous on its face as to disqualify anyone who uses it. To accept such a bizarre analogy is to show oneself incapable of serious commentary on subjects of this sort. The same could be said of the notion of “precise” placement of devices within the brain, and of the sort of speculations about the localization of depression or addiction in particular regions of the brain that people like Helen Mayberg trot out to support their baseless experimentations.
Helen Mayberg, the inimitable Helen Mayberg, is, quelle surprise, the reporter’s go-to source for confirmation of how promising this new approach is likely to be. She duly obliges, claiming that “the logic of the effort is sound and that the circuitry of this part of the brain is well-mapped compared with other regions. The key question is whether the researchers have found the precise spot for the insertion of the electrodes. The precision of where you put it is key,” she said. “Different nodes are good for different kinds of problems because circuits are abnormal in different ways.” Alcoholics, spousal abusers, criminals, you’re next! For your behaviors too, one presumes, can be solved by implanting suitable brain pacemakers. And if they don’t work? Why, the surgeon put the damned thing in the wrong place. Sorry about that. As you will note, Dr. Mayberg has thereby provided herself with a wonderful get out of jail free card when addicts don’t recover.
The surgery the Post reporter focuses on is the first of four trial cases designed to establish that “the technique is safe so that a full-scale clinical trial can be conducted.” Let us set aside the fantasy of tens of thousands of opioid addicts being subjected to seven hours of psychosurgery (a nice fantasy for medical device makers to entertain). Let us set aside the parade of serious side effects that we know those in the controlled trials of DBS for depression suffered. What justifies the experiment? The surgeon in question is quoted as acknowledging that “doctors and researchers do not yet fully understand how this works” — a statement that should more accurately read: “we have not the slightest clue of how this might work.” The prattle about dopamine that he proceeds to utter as a substitute for the scientific evidence we do not possess is an embarrassment — just speculation plucked out of thin air. As for the “inspiration” to perform the surgery, it turns out it was anecdotal reports from China! The Director of the National Institute on Drug Abuse, Nora Volkow, “spoke with a handful of the patients who underwent the procedure in China and concluded that the surgery had potential.”
This is the quality of reasoning from the person who heads this major federal agency, and that guides what dangerous interventions are allowed on an experimental basis? Words fail me.














Thank you for this important article. Grateful there is a place for you to publish it.
Report comment
“We can’t prove it but we know there must be brain problems. Where? How much? What kind?
“Who cares?
“We gotta cut or amputate something! Or at least throw some drugs at it.” 😛
Psychiatry in a nutshell.
Report comment
LOL Rachel777. The next movie out is about a couple of deranged ‘mad people’ who kidnap a few shrinks and force nothing but kindness onto them. Watch the outcome lol.
Report comment
Can’t help but wonder if this isn’t an idea that came from a Chinese student who was food poisoned and ended up being shipped off to a Mental Health facility for injection with the ‘burning flesh’ till he spilled the beans on his invention. Knowing what I know about Sun Tzu and the chapter on ‘agents’ one might send such a person that has a bad idea that sounds like a good idea and watch while they try to get it to work and fail miserably. I like the way the Chinese think 🙂
Chelmsford wasn’t that long ago either Dr Scull.
https://en.m.wikipedia.org/wiki/Chelmsford_Royal_Commission
Report comment
Thank you for the community service of articulately describing the “scientism” of psychiatry.
Report comment
If only Wilhelm Reich had come up with a device that was able to damage the human body instead of his rather innocuous orgone accumulators, maybe then the FDA wouldn’t have been able to prosecute him for fraud. His device was absolutely safe, a claim that can reliably be made neither for deep brain stimulation, nor ECT, despite industry sales blurbs to the contrary. And while I’m on the subject, psych drugs aren’t exactly sugar pills either. Deep brain stimulation has got to make you wonder, it’s so over the top! Isn’t this just another throw of the dice in pursuit of the next big therapeutic sales gimmick? Thanks for tackling the subject.
Report comment
James Hillman on the folly of reducing mind to brain:
—————————————————————-
The upshot of genetic studies leads in two (!) directions: a narrow path and a broad one. The narrow road heads toward simplistic, monogenic causes. It wants to pinpoint bits of tissue and correlate them with the vast complexity of psychic meanings. The folly of reducing mind to brain never seems to leave the Western scene. We can never give it up because it is so basic to our Western rationalist and positivist mind-set. The rationalist in the psyche wants to locate causes you can put your hands on and fix.
Machines provide the best models for meeting this desire. Take them apart, find their inner mechanisms, and then adjust their functioning by modifying their ratchets, enriching their fuel, greasing their connections. Henry Ford as father of American mental health. Result: Ritalin, Prozac, Zoloft, and dozens of other effective products for internal adjustments that we consume in abundance, millions of us, daily or twice daily. The simplistics of monogenic causes eventually leads to the control of behavior by drugs–that is, to drugged behavior.
Robert Plomin, on whose passionate, prolific, and perceptive writings this chapter has frequently relied, urgently warns against using genetics in a simplistic manner. He states: “Genetic effects on behavior are polygenic and probabilistic, not single gene and deterministic.” I gather from him a warning to psychiatry: Do not capsize your noble vessel under the weight of pharmaceutical, insurance company, and government gold, and do not set your compass toward Fantasy Island, where genetics will define “disease entities in psychiatry.” “We have learned little about the genetics of development [how genes act and interact over time] except to appreciate its complexity.” Therefore we can never arrive at that equation where one defective gene equals one clinical picture (except for true anomalies like Huntington’s chorea).
These warnings have little effect; simplistic thinking fulfills too many wishes. The heads of Henry Ford and Thomas Edison are carved into the Mount Rushmore of the mind. The monster of mechanism appears in every century of modern Western history and must be watched for by each generation–especially ours, when to hold out for “something else” besides nature or nurture means believing in ghosts or magic.
Ever since French rationalism of the seventeenth (Marin Mersenne, Nicolas de Malebranche) and eighteenth (Etienne de Condillac, Julien Offroy de La Mettrie) centuries and right through to the positivism of the nineteenth (Antoine Destutt de Tracy, Auguste Comte) in which all mental events were reduced to biology, a piece of the collective Western mind had been yolked like a dumb ox to the heavy tumbrel of French mechanistic materialism. It is astounding how people with such subtle taste as the French and with such erotic sensibility can go on and on contributing so much rationalist rigor mortis to psychology. Every import that arrives from France must be inspected for this French disease, even though it carries the fashionable label of Lacanism, Structuralism, Deconstruction, or whatever.
Today rationalism is global, computer-compatible every-where. It is the international style of the mind’s architecture. We cannot pin it to a particular flag, unless to the banners of the multinational corporation that can spend big bucks turning psychiatry, and eventually psychological thinking, and therefore soul control, toward monogenetic monotheism. One gene for one disorder: Splice the gene, teach it tricks, combine it, and the disorder is gone, or at least you don’t know you have it. The narrow path leads back to the thirties and forties of psychiatric history, though in a more refined manner and with better press releases. From 1930 into the 1950s, correlating specific brain areas with large emotional and functional concepts provided the rationale for the violence of psychosurgery and the lobotomizing of many a troubled soul at odds with circumstance.
———————————————————————————————————————
Report comment
If you are looking for more HORROR, you can look at a medical paper from 1961 on targeting the deep brain.
While today the neurosurgeon / psychiatrist is looking at the reptilian brain in the human brain or the root core of the brain, where the emotions are thought to be, back in 1961 they were destroying/ stimulating the thinking part of the brain the frontal lobe.
I did not know when lobotomy was abandoned, but from Wikipedia “Doctors in the Soviet Union concluded that the procedure was “contrary to the principles of humanity” and “‘through lobotomy’ an insane person is changed into an idiot.” By the 1970s, numerous countries had banned the procedure as had several US states.”
The medical paper of horror, https://jnnp.bmj.com/content/jnnp/24/4/353.full.pdf
or if they erase it
https://web.archive.org/web/20191115155138/https://jnnp.bmj.com/content/jnnp/24/4/353.full.pdf
Report comment
Yeah. According to Anatomy of an Epidemic, the psychiatrist who first used a drug to “treat” the “insane” praised it as a chemical lobotomy.
Now that lobotomies are no longer fashionable, but neuroleptics are most psychiatrists would like to forget that quote. Along with words like “drapetomania” and “hysterics.”
Report comment
Makes me wonder who originally had the lightbulb moment that perhaps lobotomies were not a good result? And how did that actually cause them give up those basement atrocities? Possibly, chemical lobotomies through pills came about the same time they dropped the surgeries. So it never was about the ‘inhumanity’. If it’s harmless, I’m sure the shrinks would take turns trying the new gadgets or pills on each other. Just because they don’t have the answers to what ails a human, gives them no authority to practice on them.
Report comment
Shrinks get major kickbacks from Big Pharma–often directly but always in the form of “little” gifts tallying up to $2,000-3,000 a year per company of tax free goodies.
No $$$$$$$ in lobotomies nowadays. If the APA changes its tune rest assured Big Pharma invented a fancy machine that does lazar lobotomies and is shelling out mega bucks for anyone morally bankrupt enough to shill them. Lucky for them that psychiatry exists.
Report comment
Absolutely Rachel. We don’t need horror movies. Just film pharma and medical/psychiatric practices. Watch them diagnose people with anxiety even in their last stages of disease or death. I have deduced that real crazy is far from what looks like a psychosis, rather it is methodical in nature.
Report comment
Think about the way that ECT has been repackaged to make it palatable to the general public again.
Leopard Oil (formerly known as Snake Oil) guaranteed to cure baldness, skin disorders, alcoholism, and a range of other uses.
I actually met a guy who had a lobotomy done, though he didn’t call it that. In fact I don’t think he knew much about anything tbh lol. “Medication time, medication time”.
Report comment
But realistically they are trying to ‘help’ people. Such an obsessed people, randomly sticking things into people’s brains. Like a Frankenstein movie. They would not do it to their own dog. Hopefully one day they succeed in making everyone normal and be just like them. We can sit around and discuss each other and do surveys, instead of treating each other, Lord knows no shrink will ever get something put in his brain or voluntarily take ‘serotonin’. He took the test, his levels are all good thank you very much.
Report comment
“When comparisons are made between studies of drugs sponsored by Big Pharma, and trials sponsored by federal dollars, those with drug company sponsorship routinely report far more favorable results than studies where no commercial interests are at stake.” How shocking (sarcasm).
Big Pharma trials of their own drugs should never have been made legal, dah. But I do think all money supposedly used for trials by big Pharma, should be given to the governments of our world, and many other independent researchers. And not just the ones who work for the “elite” universities, who’ve already proven their employees have “delusions of grandeur” they’re “second only to God.” (Joseph Beiderman story …)
“The situation is even worse when it comes to medical devices.” I have no doubt, since my parents’ next door neighbors were makers of medical devices, they were constantly doing renovations on their large home, and they were able to donate many millions to our local hospitals. I’m quite certain there are lack of ethics problems with the makers of medical devices and the hospitals receiving these funds.
“The etiological understanding of mental disorders remains strikingly primitive, and such treatments as we have provide at best a measure of symptomatic relief to a subset of these patients, often at a very heavy price in terms of side effects.”
Actually, the “etiological understanding of mental disorders,” at least of the two most “serious mental health disorders,” has already been pointed out by the non-mental health employed, independent scientific psychopharmacology researchers. The etiology of most “bipolar” is likely the adverse effects of the ADHD drugs and antidepressants being misdiagnosed as “bipolar,” as Robert Whitaker pointed out in “Anatomy …”
https://www.alternet.org/2010/04/are_prozac_and_other_psychiatric_drugs_causing_the_astonishing_rise_of_mental_illness_in_america/
And this disclaimer was unethically taken out of the DSM-IV-TR, by the writers of the DSM5, likely to spite Robert Whitaker.
“Note: Manic-like episodes that are clearly caused by somatic antidepressant treatment (e.g., medication, electroconvulsive therapy, light therapy) should not count toward a diagnosis of Bipolar I Disorder.”
And the etiology of most “schizophrenia” is likely the fact that the antipsychotics/neuroleptics can create the negative symptoms of “schizophrenia,” via neuroleptic induced deficit syndrome. And the antipsychotics/neuroleptics can create the positive symptoms of “schizophrenia,” like psychosis and hallucinations, via antidepressant and/or antipsychotic induced anticholinergic toxidrome.
https://en.wikipedia.org/wiki/Neuroleptic-induced_deficit_syndrome
https://en.wikipedia.org/wiki/Toxidrome
I agree, the truth of the systemic crimes of the “mental health” industry need to be made known. And their continued desire to experiment on the most vulnerable, is morally repugnant. Psychiatry has killed too many already.
https://breggin.com/wp-content/uploads/2008/01/psychiatrysrole.pbreggin.1993.pdf
https://www.naturalnews.com/049860_psych_drugs_medical_holocaust_Big_Pharma.html
Report comment
Thank you Dr. Scull for reporting on this. As Diana noted above I’m glad there is a website to publish this info for a large audience. This corruption and greed is despicable.
As you noted “Mental patients remain extremely vulnerable” – yet people deemed to be “mental patients” are continually used as guinea pigs. Nothing has changed from the days of lobotomy.
And…. “In other words, in nearly two-thirds of cases, these physicians lied, and of course they did so with impunity. So it is that “evidence-based medicine” becomes “evidence-biased medicine”.
The darkest and most disgusting reality I have come to realize from my own psychiatric encounter is – that physicians, especially psychiatrists, lie with impunity and inflict great harm with impunity.
Report comment
In case it works, it is simply due to the “Placebo effect” – expectations can play a large role in healing.
Report comment
Wouldn’t it be easier to just build the brain in the laboratory, first? This reads like the story of mad scientists gone awry. Mary Shelley was foretelling.
Report comment
LOL @Alex. I enjoyed that comment.
Report comment