Tag: financial conflicts of interest
Antidepressant Trials “Hijacked for Marketing Purposes,” Researchers Say
About half of the large antidepressant trials are biased enough to be considered “seeding trials,” according to the researchers.
Undisclosed Financial Conflicts of Interest in the DSM-5: An Interview with...
On the Mad in America podcast we talk with Lisa Cosgrove and Brian Piper about their BMJ paper entitled "Undisclosed Financial Conflicts of Interest in the DSM-5 TR: Cross-Sectional Analysis"
Screening for Bipolar: Have You Ever Been “Unusually Happy” for More...
A new questionnaire funded by AbbVie conflates antidepressant side effects with bipolar disorder and doesn’t actually meet the criteria for being considered “screening.”
Patients’ Drug Options Under Medicaid Influenced by Drugmakers
From NPR: "A Center for Public Integrity and NPR investigation found drug companies have infiltrated nearly every part of the process that determines how their drugs will be covered...
Where Did the Guidelines Go?
From The Lown Institute: "For twenty years, the Agency for Healthcare Research and Quality maintained a digital archive of medical guidelines as a resource for...
How a Flood of Corporate Funding Can Distort NIH Research
From The Washington Post: "At the heart of the matter is money. As Congress has declined to spend more on research, many academics have been...
Researchers Challenge Industry-Friendly Depression Guideline
Review of a new mixed depression guideline reveals financial bias of guideline developers and lack of evidence supporting recommendations for prescribing of antipsychotics.
Pharmaceutical Industry Gifts Linked to Costlier Prescribing Practices
New study finds that pharmaceutical industry gifts to medical providers in Washington D.C. are associated with higher cost, higher volume prescribing behavior.
Opioid Conflict-of-Interest Reveals Big Pharma’s Ties to Doctors
From CBC News: A McMaster University committee has been assigned to develop new opioid-prescribing guidelines for Canada's doctors. While the rules explicitly prohibited anyone with ties...
Majority of Pediatric Antidepressant Industry Trials Considered Low Quality
Meta-analyses including studies that detail these trials could be presenting misleading information.
Conflicts of Interest Found in Psychotherapy Research
Research highlights the need for conflict of interest transparency and management in systemic reviews of psychological therapies.
“21st Century Cures Bill Would Weaken Requirements for Disclosing Industry Ties”
Ed Silverman reports for STAT that a provision tucked into the 21st Century Cares legislation exempts companies from reporting payments made to doctors, journals,...
Study 329 Taper Phase
Most doctors still affect surprise at the idea SSRIs might come with withdrawal problems. Regulators knew very clearly since 2002 about the problems, but have decided to leave any communication of these issues in company hands.
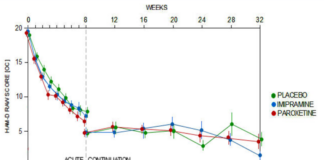
Study 329 Continuation Phase
All the fuss about Study 329 centers on its 8-week acute phase. But this study had a 24-week Continuation Phase that has never been published. Until Now.
Restoring Study 329: Letter to BMJ
When we set out to restore GSK’s misreported Study 329 of paroxetine for adolescent depression under the RIAT initiative, we had no idea of the magnitude of the task we were undertaking. After almost a year, we were relieved to finally complete a draft and submit it to the BMJ, who had earlier indicated an interest in publishing our restoration. But that was the beginning of another year of peer review that we believed went beyond enhancing our paper and became rather an interrogation of our honesty and integrity. Frankly, we were offended that our work was subject to such checks when papers submitted by pharmaceutical companies with fraud convictions are not.
“Financial Conflicts of Interest in Medicine”
Citing the work of Lisa Cosgrove and Robert Whitaker in Psychiatry Under the Influence, Giovanni A. Fava, MD, provides an analysis of some subtle...
Poor Evidence and Substantial Bias in Ritalin Studies
The authors of a large scale well-conducted systematic review of methylphenidate, also known as Ritalin, conclude that there is a lack of quality evidence for the drug’s effectiveness. Their research also revealed that Ritalin can cause sleep problems and decreased appetite in children.
Study 329: 50 Shades of Gray
Access to data is more important than access to information about conflicts of interest. It is only when there is access to the data that we can see if interests are conflicting and take that into account. Problems don’t get solved unless someone is motivated for some reason. We need the bias that pharmaceutical companies bring to bear in their defense of a product, along with the bias of those who might have been injured by a treatment. Both of these biases can distort the picture but it’s when people with differing points of view agree on what is right in front of their noses that we can begin to have some confidence about what we have.
Nominee to Lead FDA Removed Name From Recent Publications
Sheila Kaplan for the Boston Globe reports that Dr. Robert Califf, the Obama administration's nominee to lead the Food and Drug Administration (FDA), has removed his name from a series of scientific papers that he recently coauthored. The decision to remove his name, against publication ethics standards, has brought Califf under renewed criticism.